Gregory, G. A. et al. Global incidence, prevalence, and mortality of type 1 diabetes in 2021 with projection to 2040: a modelling study. Lancet Diabetes Endocrinol. 10, 741–760 (2022).
Google Scholar
Herold, K. C. et al. An anti-CD3 antibody, teplizumab, in relatives at risk for type 1 diabetes. N. Engl. J. Med. 381, 603–613 (2019).
Google Scholar
Gillespie, K. M. Type 1 diabetes: pathogenesis and prevention. Can. Med. Assoc. J. 175, 165–170 (2006).
Google Scholar
Dayan, C. M., Korah, M., Tatovic, D., Bundy, B. N. & Herold, K. C. Changing the landscape for type 1 diabetes: the first step to prevention. Lancet 394, 1286–1296 (2019).
Google Scholar
McKnight, J. A. et al. Glycaemic control of type 1 diabetes in clinical practice early in the 21st century: an international comparison. Diabet. Med. 32, 1036–1050 (2015).
Google Scholar
Ogle, G. D. et al. Global estimates of incidence of type 1 diabetes in children and adolescents: results from the International Diabetes Federation Atlas, 10th edition. Diabetes Res. Clin. Pract. 183, 109083 (2022).
Google Scholar
Speake, C. & Greenbaum, C. J. Approval of teplizumab: implications for patients. Nat. Rev. Endocrinol. 19, 377–378 (2023).
Google Scholar
Forbes, J. M. et al. Advanced glycation end products as predictors of renal function in youth with type 1 diabetes. Sci. Rep. 11, 9422 (2021).
Google Scholar
Le Bagge, S., Fotheringham, A. K., Leung, S. S. & Forbes, J. M. Targeting the receptor for advanced glycation end products (RAGE) in type 1 diabetes. Med. Res. Rev. 40, 1200–1219 (2020).
Google Scholar
Leung, S. S. et al. Soluble RAGE prevents type 1 diabetes expanding functional regulatory T cells. Diabetes 71, 1994–2008 (2022).
Google Scholar
Chhipa, A. S., Borse, S. P., Baksi, R., Lalotra, S. & Nivsarkar, M. Targeting receptors of advanced glycation end products (RAGE): preventing diabetes induced cancer and diabetic complications. Pathol. Res. Pract. 215, 152643 (2019).
Google Scholar
Magliano, D. J. & Boyko, E. J. IDF Diabetes Atlas (International Diabetes Federation, 2021).
Ogle, G. D., Middlehurst, A. C. & Silink, M. The IDF life for a child program index of diabetes care for children and youth. Pediatr. Diabetes 17, 374–384 (2016).
Google Scholar
American Diabetes Association Professional Practice Committee 2. Diagnosis and classification of diabetes: standards of care in diabetes – 2024. Diabetes Care 47, S20–S42 (2024).
Google Scholar
JDRF Australia. Economic cost of type 1 diabetes in Australia. Accenture (2021).
Joish, V. N. et al. Estimation of annual health care costs for adults with type 1 diabetes in the United States. J. Manag. Care Spec. Pharm. 26, 311–318 (2020).
Google Scholar
Rawshani, A. et al. Excess mortality and cardiovascular disease in young adults with type 1 diabetes in relation to age at onset: a nationwide, register-based cohort study. Lancet 392, 477–486 (2018).
Google Scholar
Livingstone, S. J. et al. Estimated life expectancy in a Scottish cohort with type 1 diabetes, 2008-2010. JAMA 313, 37–44 (2015).
Google Scholar
Rewers, M. & Ludvigsson, J. Environmental risk factors for type 1 diabetes. Lancet 387, 2340–2348 (2016).
Google Scholar
Knip, M. & Simell, O. Environmental triggers of type 1 diabetes. Cold Spring Harb. Perspect. Med. 2, a007690 (2012).
Google Scholar
Hagopian, W. A. et al. The environmental determinants of diabetes in the young (TEDDY): genetic criteria and international diabetes risk screening of 421 000 infants. Pediatr. Diabetes 12, 733–743 (2011).
Google Scholar
TEDDY study group The environmental determinants of diabetes in the young (TEDDY) study. Ann. N. Y. Acad. Sci. 1150, 1–13 (2008).
Google Scholar
Eringsmark Regnéll, S. & Lernmark, A. The environment and the origins of islet autoimmunity and type 1 diabetes. Diabet. Med. 30, 155–160 (2013).
Google Scholar
Noble, J. A. & Valdes, A. M. Genetics of the HLA region in the prediction of type 1 diabetes. Curr. Diab Rep. 11, 533–542 (2011).
Google Scholar
Turtinen, M., Härkönen, T., Parkkola, A., Ilonen, J. & Knip, M. Characteristics of familial type 1 diabetes: effects of the relationship to the affected family member on phenotype and genotype at diagnosis. Diabetologia 62, 2025–2039 (2019).
Google Scholar
Sharp, S. A. et al. Development and standardization of an improved type 1 diabetes genetic risk score for use in newborn screening and incident diagnosis. Diabetes Care 42, 200–207 (2019).
Google Scholar
Mameli, C. et al. Lessons and gaps in the prediction and prevention of type 1 diabetes. Pharmacol. Res. 193, 106792 (2023).
Google Scholar
Bottazzo, G. F., Florin-Christensen, A. & Doniach, D. Islet-cell antibodies in diabetes mellitus with autoimmune polyendocrine deficiencies. Lancet 2, 1279–1283 (1974).
Google Scholar
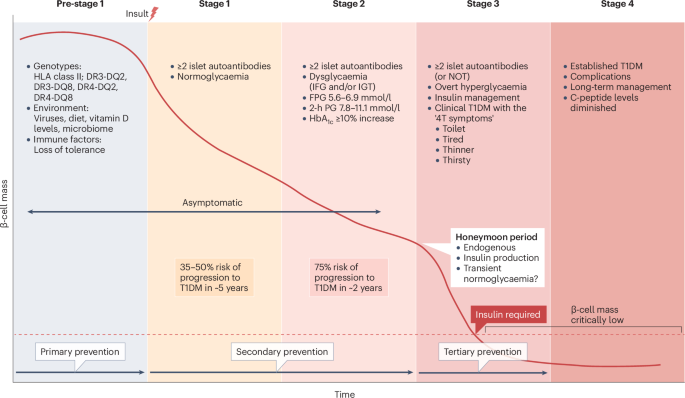
Insel, R. A. et al. Staging presymptomatic type 1 diabetes: a scientific statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care 38, 1964–1974 (2015).
Google Scholar
Ziegler, A. G. & Nepom, G. T. Prediction and pathogenesis in type 1 diabetes. Immunity 32, 468–478 (2010).
Google Scholar
Atkinson, M. A. & Mirmira, R. G. The pathogenic “symphony” in type 1 diabetes: a disorder of the immune system, β cells, and exocrine pancreas. Cell Metab. 35, 1500–1518 (2023).
Google Scholar
Ziegler, A. G. et al. Seroconversion to multiple islet autoantibodies and risk of progression to diabetes in children. JAMA 309, 2473–2479 (2013).
Google Scholar
Krischer, J. P. et al. Predictors of the initiation of islet autoimmunity and progression to multiple autoantibodies and clinical diabetes: the TEDDY study. Diabetes Care 45, 2271–2281 (2022).
Google Scholar
Hummel, S. & Ziegler, A. G. Early determinants of type 1 diabetes: experience from the BABYDIAB and BABYDIET studies. Am. J. Clin. Nutr. 94, 1821s–1823s (2011).
Google Scholar
Phillip, M. et al. Consensus guidance for monitoring individuals with islet autoantibody-positive pre-stage 3 type 1 diabetes. Diabetes Care 47, 1276–1298 (2024).
Google Scholar
Jacobsen, L. M. et al. The risk of progression to type 1 diabetes is highly variable in individuals with multiple autoantibodies following screening. Diabetologia 63, 588–596 (2020).
Google Scholar
Patel, S. K., Ma, C. S., Fourlanos, S. & Greenfield, J. R. Autoantibody-negative type 1 diabetes: a neglected subtype. Trends Endocrinol. Metab. 32, 295–305 (2021).
Google Scholar
Hanna, S. J. et al. Slow progressors to type 1 diabetes lose islet autoantibodies over time, have few islet antigen-specific CD8+ T cells and exhibit a distinct CD95hi B cell phenotype. Diabetologia 63, 1174–1185 (2020).
Google Scholar
Carlsson, A. et al. Absence of islet autoantibodies and modestly raised glucose values at diabetes diagnosis should lead to testing for MODY: lessons from a 5-year pediatric Swedish national cohort study. Diabetes Care 43, 82–89 (2020).
Google Scholar
Holt, R. I. G. et al. The management of type 1 diabetes in adults. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 64, 2609–2652 (2021).
Google Scholar
Ogle, G. D., Wang, F., Gregory, G. A. & Maniam, J. Type 1 diabetes estimates in children and adults. International Diabetes Federation diabetesatlas.org/media/uploads/sites/3/2025/03/IDF-T1D-Index-Report.pdf (2022).
Herzog, K. et al. Alterations in biomarkers related to glycemia, lipid metabolism, and inflammation up to 20 years before diagnosis of type 1 diabetes in adults: findings from the AMORIS cohort. Diabetes Care 45, 330–338 (2021).
Google Scholar
Thomas, N. J. et al. Age of diagnosis does not alter the presentation or progression of robustly defined adult-onset type 1 diabetes. Diabetes Care 46, 1156–1163 (2023).
Google Scholar
Bluestone, J. A., Herold, K. & Eisenbarth, G. Genetics, pathogenesis and clinical interventions in type 1 diabetes. Nature 464, 1293–1300 (2010).
Google Scholar
Gepts, W. & Lecompte, P. M. The pancreatic islets in diabetes. Am. J. Med. 70, 105–115 (1981).
Google Scholar
Foulis, A. K., Liddle, C. N., Farquharson, M. A., Richmond, J. A. & Weir, R. S. The histopathology of the pancreas in type I (insulin-dependent) diabetes mellitus: a 25-year review of deaths in patients under 20 years of age in the United Kingdom. Diabetologia 29, 267–274 (1986).
Google Scholar
Bottazzo, G. F. Lawrence lecture. death of a beta cell: homicide or suicide? Diabet. Med. 3, 119–130 (1986).
Google Scholar
Atkinson, M. A. et al. How does type 1 diabetes develop?: the notion of homicide or β-cell suicide revisited. Diabetes 60, 1370–1379 (2011).
Google Scholar
Roep, B. O., Thomaidou, S., van Tienhoven, R. & Zaldumbide, A. Type 1 diabetes mellitus as a disease of the β-cell (do not blame the immune system?). Nat. Rev. Endocrinol. 17, 150–161 (2021).
Google Scholar
Wong, S. et al. Expression of the co-stimulator molecule B7-1 in pancreatic beta-cells accelerates diabetes in the NOD mouse. Diabetes 44, 326–329 (1995).
Google Scholar
Wong, F. S. et al. Identification of an MHC class I-restricted autoantigen in type 1 diabetes by screening an organ-specific cDNA library. Nat. Med. 5, 1026–1031 (1999).
Google Scholar
Klein, D. et al. A functional CD40 receptor is expressed in pancreatic beta cells. Diabetologia 48, 268–276 (2005).
Google Scholar
Narendran, P., Mannering, S. I. & Harrison, L. C. Proinsulin – a pathogenic autoantigen in type 1 diabetes. Autoimmun. Rev. 2, 204–210 (2003).
Google Scholar
Delong, T. et al. Diabetogenic T-cell clones recognize an altered peptide of chromogranin A. Diabetes 61, 3239–3246 (2012).
Google Scholar
Delong, T. et al. Pathogenic CD4 T cells in type 1 diabetes recognize epitopes formed by peptide fusion. Science 351, 711–714 (2016).
Google Scholar
Gottlieb, P. A. et al. Chromogranin A is a T cell antigen in human type 1 diabetes. J. Autoimmun. 50, 38–41 (2014).
Google Scholar
Mannering, S. I., Rubin, A. F., Wang, R. & Bhattacharjee, P. Identifying new hybrid insulin peptides (HIPs) in type 1 diabetes. Front. Immunol. 12, 667870 (2021).
Google Scholar
Baekkeskov, S. et al. Expression of major histocompatibility antigens on pancreatic islet cells. Proc. Natl Acad. Sci. 78, 6456–6460 (1981).
Google Scholar
Foulis, A. K., Farquharson, M. A. & Hardman, R. Aberrant expression of class II major histocompatibility complex molecules by B cells and hyperexpression of class I major histocompatibility complex molecules by insulin containing islets in type 1 (insulin-dependent) diabetes mellitus. Diabetologia 30, 333–343 (1987).
Google Scholar
Richardson, S. J. et al. Islet cell hyperexpression of HLA class I antigens: a defining feature in type 1 diabetes. Diabetologia 59, 2448–2458 (2016).
Google Scholar
Russell, M. A. et al. HLA class II antigen processing and presentation pathway components demonstrated by transcriptome and protein analyses of islet β-cells from donors with type 1 diabetes. Diabetes 68, 988–1001 (2019).
Google Scholar
Itoh, N. et al. Mononuclear cell infiltration and its relation to the expression of major histocompatibility complex antigens and adhesion molecules in pancreas biopsy specimens from newly diagnosed insulin-dependent diabetes mellitus patients. J. Clin. Investig. 92, 2313–2322 (1993).
Google Scholar
Unger, W. W. et al. Islet-specific CTL cloned from a type 1 diabetes patient cause beta-cell destruction after engraftment into HLA-A2 transgenic NOD/scid/IL2RG null mice. PLoS ONE 7, e49213 (2012).
Google Scholar
von Herrath, M. G., Guerder, S., Lewicki, H., Flavell, R. A. & Oldstone, M. B. Coexpression of B7-1 and viral (“self”) transgenes in pancreatic beta cells can break peripheral ignorance and lead to spontaneous autoimmune diabetes. Immunity 3, 727–738 (1995).
Google Scholar
von Herrath, M. G., Holz, A., Homann, D. & Oldstone, M. B. Role of viruses in type I diabetes. Semin. Immunol. 10, 87–100 (1998).
Google Scholar
Dahl-Jørgensen, K. Virus as the cause of type 1 diabetes. Trends Mol. Med. 30, 1020–1027 (2024).
Google Scholar
Coppieters, K. T., Boettler, T. & von Herrath, M. Virus infections in type 1 diabetes. Cold Spring Harb. Perspect. Med. 2, a007682 (2012).
Google Scholar
Kuric, E. et al. Demonstration of tissue resident memory CD8 T cells in insulitic lesions in adult patients with recent-onset type 1 diabetes. Am. J. Pathol. 187, 581–588 (2017).
Google Scholar
Gagnerault, M. C., Luan, J. J., Lotton, C. & Lepault, F. Pancreatic lymph nodes are required for priming of beta cell reactive T cells in NOD mice. J. Exp. Med. 196, 369–377 (2002).
Google Scholar
Kurts, C. et al. Constitutive class I-restricted exogenous presentation of self antigens in vivo. J. Exp. Med. 184, 923–930 (1996).
Google Scholar
Calderon, B., Suri, A. & Unanue, E. R. In CD4+ T-cell-induced diabetes, macrophages are the final effector cells that mediate islet beta-cell killing: studies from an acute model. Am. J. Pathol. 169, 2137–2147 (2006).
Google Scholar
Sandor, A. M., Jacobelli, J. & Friedman, R. S. Immune cell trafficking to the islets during type 1 diabetes. Clin. Exp. Immunol. 198, 314–325 (2019).
Google Scholar
Hull, C. M., Peakman, M. & Tree, T. I. M. Regulatory T cell dysfunction in type 1 diabetes: what’s broken and how can we fix it? Diabetologia 60, 1839–1850 (2017).
Google Scholar
Buckner, J. H. Mechanisms of impaired regulation by CD4+CD25+FOXP3+ regulatory T cells in human autoimmune diseases. Nat. Rev. Immunol. 10, 849–859 (2010).
Google Scholar
Leete, P. et al. Differential insulitic profiles determine the extent of β-cell destruction and the age at onset of type 1 diabetes. Diabetes 65, 1362–1369 (2016).
Google Scholar
Willcox, A., Richardson, S. J., Bone, A. J., Foulis, A. K. & Morgan, N. G. Analysis of islet inflammation in human type 1 diabetes. Clin. Exp. Immunol. 155, 173–181 (2009).
Google Scholar
Redondo, M. J. & Morgan, N. G. Heterogeneity and endotypes in type 1 diabetes mellitus. Nat. Rev. Endocrinol. 19, 542–554 (2023).
Google Scholar
Smith, M. J., Cambier, J. C. & Gottlieb, P. A. Endotypes in T1D: B lymphocytes and early onset. Curr. Opin. Endocrinol. Diabetes Obes. 27, 225–230 (2020).
Google Scholar
Vandamme, C. & Kinnunen, T. B cell helper T cells and type 1 diabetes. Scand. J. Immunol. 92, e12943 (2020).
Google Scholar
Hanley, P. et al. Circulating B cells in type 1 diabetics exhibit fewer maturation-associated phenotypes. Clin. Immunol. 183, 336–343 (2017).
Google Scholar
Mallone, R. & Brezar, V. To B or not to B: (anti)bodies of evidence on the crime scene of type 1 diabetes? Diabetes 60, 2020–2022 (2011).
Google Scholar
Pinto, A. I., Smith, J., Kissack, M. R., Hogg, K. G. & Green, E. A. Thymic B cell-mediated attack of thymic stroma precedes type 1 diabetes development. Front. Immunol. 9, 1281 (2018).
Google Scholar
Karges, B. et al. A comparison of familial and sporadic type 1 diabetes among young patients. Diabetes Care 44, 1116–1124 (2021).
Google Scholar
Skyler, J. S. Primary and secondary prevention of type 1 diabetes. Diabet. Med. 30, 161–169 (2013).
Google Scholar
Couper, J. J. et al. ISPAD clinical practice consensus guidelines 2018: stages of type 1 diabetes in children and adolescents. Pediatr. Diabetes 19, 20–27 (2018).
Google Scholar
Hu, Z. D. & Deng, A. M. Autoantibodies in pre-clinical autoimmune disease. Clin. Chim. Acta 437, 14–18 (2014).
Google Scholar
Oram, R. A., Sims, E. K. & Evans-Molina, C. Beta cells in type 1 diabetes: mass and function; sleeping or dead? Diabetologia 62, 567–577 (2019).
Google Scholar
Palmer, J. P. et al. C-peptide is the appropriate outcome measure for type 1 diabetes clinical trials to preserve beta-cell function: report of an ADA workshop, 21–22 October 2001. Diabetes 53, 250–264 (2004).
Google Scholar
Skyler, J. S. Immune intervention studies in insulin-dependent diabetes mellitus. Diabetes Metab. Rev. 3, 1017–1035 (1987).
Google Scholar
Stiller, C. R. et al. Effects of cyclosporine immunosuppression in insulin-dependent diabetes mellitus of recent onset. Science 223, 1362–1367 (1984).
Google Scholar
Feutren, G. et al. Cyclosporin increases the rate and length of remissions in insulin-dependent diabetes of recent onset. Results of a multicentre double-blind trial. Lancet 2, 119–124 (1986).
Google Scholar
Parving, H. H. et al. Cyclosporine nephrotoxicity in type 1 diabetic patients. A 7-year follow-up study. Diabetes Care 22, 478–483 (1999).
Google Scholar
Chatenoud, L., Thervet, E., Primo, J. & Bach, J. F. Anti-CD3 antibody induces long-term remission of overt autoimmunity in nonobese diabetic mice. Proc. Natl Acad. Sci. USA 91, 123–127 (1994).
Google Scholar
Nicoletti, F. et al. Prevention of cyclophosphamide-induced diabetes in the NOD/WEHI mouse with deoxyspergualin. Clin. Exp. Immunol. 91, 232–236 (1993).
Google Scholar
Gaglia, J. & Kissler, S. Anti-CD3 antibody for the prevention of type 1 diabetes: a story of perseverance. Biochemistry 58, 4107–4111 (2019).
Google Scholar
Herold, K. C. et al. Treatment of patients with new onset type 1 diabetes with a single course of anti-CD3 mAb teplizumab preserves insulin production for up to 5 years. Clin. Immunol. 132, 166–173 (2009).
Google Scholar
Herold, K. C. et al. Anti-CD3 monoclonal antibody in new-onset type 1 diabetes mellitus. N. Engl. J. Med. 346, 1692–1698 (2002).
Google Scholar
Keymeulen, B. et al. Insulin needs after CD3-antibody therapy in new-onset type 1 diabetes. N. Engl. J. Med. 352, 2598–2608 (2005).
Google Scholar
Keymeulen, B. et al. Four-year metabolic outcome of a randomised controlled CD3-antibody trial in recent-onset type 1 diabetic patients depends on their age and baseline residual beta cell mass. Diabetologia 53, 614–623 (2010).
Google Scholar
Hagopian, W. et al. Teplizumab preserves C-peptide in recent-onset type 1 diabetes: two-year results from the randomized, placebo-controlled Protégé trial. Diabetes 62, 3901–3908 (2013).
Google Scholar
Ambery, P. et al. Efficacy and safety of low-dose otelixizumab anti-CD3 monoclonal antibody in preserving C-peptide secretion in adolescent type 1 diabetes: DEFEND-2, a randomized, placebo-controlled, double-blind, multi-centre study. Diabet. Med. 31, 399–402 (2014).
Google Scholar
Aronson, R. et al. Low-dose otelixizumab anti-CD3 monoclonal antibody DEFEND-1 study: results of the randomized phase III study in recent-onset human type 1 diabetes. Diabetes Care 37, 2746–2754 (2014).
Google Scholar
Sherry, N. et al. Teplizumab for treatment of type 1 diabetes (Protege study): 1-year results from a randomised, placebo-controlled trial. Lancet 378, 487–497 (2011).
Google Scholar
Evans-Molina, C. & Oram, R. A. Teplizumab approval for type 1 diabetes in the USA. Lancet Diabetes Endocrinol. 11, 76–77 (2023).
Google Scholar
Sims, E. K. et al. Teplizumab improves and stabilizes beta cell function in antibody-positive high-risk individuals. Sci. Transl. Med. 13, eabc8980 (2021).
Google Scholar
Ramos, E. L. et al. Teplizumab and β-cell function in newly diagnosed type 1 diabetes. N. Engl. J. Med. 389, 2151–2161 (2023).
Google Scholar
Keymeulen, B. et al. Transient Epstein-Barr virus reactivation in CD3 monoclonal antibody-treated patients. Blood 115, 1145–1155 (2010).
Google Scholar
Hummel, S. et al. Children diagnosed with presymptomatic type 1 diabetes through public health screening have milder diabetes at clinical manifestation. Diabetologia 66, 1633–1642 (2023).
Google Scholar
Fredheim, S. et al. Diabetic ketoacidosis at the onset of type 1 diabetes is associated with future HbA1c levels. Diabetologia 56, 995–1003 (2013).
Google Scholar
Duca, L. M. et al. Diabetic ketoacidosis at diagnosis of type 1 diabetes and glycemic control over time: the SEARCH for diabetes in youth study. Pediatr. Diabetes 20, 172–179 (2019).
Google Scholar
Duca, L. M., Wang, B., Rewers, M. & Rewers, A. Diabetic ketoacidosis at diagnosis of type 1 diabetes predicts poor long-term glycemic control. Diabetes Care 40, 1249–1255 (2017).
Google Scholar
Cameron, F. J. et al. Neurological consequences of diabetic ketoacidosis at initial presentation of type 1 diabetes in a prospective cohort study of children. Diabetes Care 37, 1554–1562 (2014).
Google Scholar
Whittemore, R., Jaser, S., Chao, A., Jang, M. & Grey, M. Psychological experience of parents of children with type 1 diabetes: a systematic mixed-studies review. Diabetes Educ. 38, 562–579 (2012).
Google Scholar
Park, H., Adsit, F. G. & Boyington, J. C. The 1.5 A crystal structure of human receptor for advanced glycation endproducts (RAGE) ectodomains reveals unique features determining ligand binding. J. Biol. Chem. 285, 40762–40770 (2010).
Google Scholar
Hofmann, M. A. et al. RAGE mediates a novel proinflammatory axis: a central cell surface receptor for S100/calgranulin polypeptides. Cell 97, 889–901 (1999).
Google Scholar
Hori, O. et al. The receptor for advanced glycation end products (RAGE) is a cellular binding site for amphoterin. Mediation of neurite outgrowth and co-expression of rage and amphoterin in the developing nervous system. J. Biol. Chem. 270, 25752–25761 (1995).
Google Scholar
Yan, S. D. et al. RAGE and amyloid-β peptide neurotoxicity in Alzheimer’s disease. Nature 382, 685–691 (1996).
Google Scholar
Sousa, M. M., Yan, S. D., Stern, D. & Saraiva, M. J. Interaction of the receptor for advanced glycation end products (RAGE) with transthyretin triggers nuclear transcription factor kB (NF-kB) activation. Lab. Invest. 80, 1101–1110 (2000).
Google Scholar
Chavakis, T. et al. The pattern recognition receptor (RAGE) is a counterreceptor for leukocyte integrins: a novel pathway for inflammatory cell recruitment. J. Exp. Med. 198, 1507–1515 (2003).
Google Scholar
Ma, W. et al. RAGE binds C1q and enhances C1q-mediated phagocytosis. Cell Immunol. 274, 72–82 (2012).
Google Scholar
Taguchi, A. et al. Blockade of RAGE-amphoterin signalling suppresses tumour growth and metastases. Nature 405, 354–360 (2000).
Google Scholar
Yeh, C. H. et al. Requirement for p38 and p44/p42 mitogen-activated protein kinases in RAGE-mediated nuclear factor-κB transcriptional activation and cytokine secretion. Diabetes 50, 1495–1504 (2001).
Google Scholar
Grimm, S. et al. Advanced-glycation-end-product-induced formation of immunoproteasomes: involvement of RAGE and Jak2/STAT1. Biochem. J. 448, 127–139 (2012).
Google Scholar
Huttunen, H. J., Fages, C. & Rauvala, H. Receptor for advanced glycation end products (RAGE)-mediated neurite outgrowth and activation of NF-κB require the cytoplasmic domain of the receptor but different downstream signaling pathways. J. Biol. Chem. 274, 19919–19924 (1999).
Google Scholar
Ott, C. et al. Role of advanced glycation end products in cellular signaling. Redox Biol. 2, 411–429 (2014).
Google Scholar
Xie, J., Mendez, J. D., Mendez-Valenzuela, V. & Aguilar-Hernandez, M. M. Cellular signalling of the receptor for advanced glycation end products (RAGE). Cell Signal. 25, 2185–2197 (2013).
Google Scholar
Du, C. et al. Advanced glycation end products and inflammation in type 1 diabetes development. Cells 11, 3503 (2022).
Google Scholar
Sakaguchi, M. et al. Signal diversity of receptor for advanced glycation end products. Acta Med. Okayama 71, 459–465 (2017).
Google Scholar
Leung, S. S., Forbes, J. M. & Borg, D. J. Receptor for advanced glycation end products (RAGE) in type 1 diabetes pathogenesis. Curr. Diab Rep. 16, 100 (2016).
Google Scholar
Leung, S. S. et al. Alpha cell receptor for advanced glycation end products associate with glucagon expression in type 1 diabetes. Sci. Rep. 13, 12948 (2023).
Google Scholar
Durning, S. P. et al. The receptor for advanced glycation endproducts drives T cell survival and inflammation in type 1 diabetes mellitus. J. Immunol. 197, 3076–3085 (2016).
Google Scholar
Akirav, E. M. et al. RAGE expression in human T cells: a link between environmental factors and adaptive immune responses. PLoS ONE 7, e34698 (2012).
Google Scholar
Chen, Y. et al. RAGE ligation affects T cell activation and controls T cell differentiation. J. Immunol. 181, 4272–4278 (2008).
Google Scholar
Akirav, E. M. et al. The receptor for advanced glycation end products (RAGE) affects T cell differentiation in OVA induced asthma. PLoS ONE 9, e95678 (2014).
Google Scholar
Moser, B. et al. Receptor for advanced glycation end products expression on T cells contributes to antigen-specific cellular expansion in vivo. J. Immunol. 179, 8051–8058 (2007).
Google Scholar
Coughlan, M. T. et al. Advanced glycation end products are direct modulators of β-cell function. Diabetes 60, 2523–2532 (2011).
Google Scholar
Joshi, A. A. et al. RAGE antagonism with azeliragon improves xenograft rejection by T cells in humanized mice. Clin. Immunol. 245, 109165 (2022).
Google Scholar
Burstein, A. H., Dunn, I., Gooch, A. M. & Valcarce, C. Effects of azeliragon on ADAS-cog and CDR domains and individual items in patients with mild Alzheimer’s disease and type 2 diabetes (T2D). Alzheimer’s Dement. 16, e041198 (2020).
Google Scholar
Burstein, A. H. et al. Effect of TTP488 in patients with mild to moderate Alzheimer’s disease. BMC Neurol. 14, 12 (2014).
Google Scholar
Burstein, A. H. et al. Development of azeliragon, an oral small molecule antagonist of the receptor for advanced glycation endproducts, for the potential slowing of loss of cognition in mild Alzheimer’s disease. J. Prev. Alzheimers Dis. 5, 149–154 (2018).
Google Scholar
Manfredi, A. A. et al. Maturing dendritic cells depend on RAGE for in vivo homing to lymph nodes. J. Immunol. 180, 2270–2275 (2008).
Google Scholar
Wittkowski, H. et al. Acute Kawasaki disease is associated with reverse regulation of soluble receptor for advance glycation end products and its proinflammatory ligand S100A12. Arthritis Rheum. 56, 4174–4181 (2007).
Google Scholar
Koenig, R. J. et al. Correlation of glucose regulation and hemoglobin A1c in diabetes mellitus. N. Engl. J. Med. 295, 417–420 (1976).
Google Scholar
Baynes, J. W. The role of AGEs in aging: causation or correlation. Exp. Gerontol. 36, 1527–1537 (2001).
Google Scholar
Zhao, Z. et al. Advanced glycation end products inhibit glucose-stimulated insulin secretion through nitric oxide-dependent inhibition of cytochrome c oxidase and adenosine triphosphate synthesis. Endocrinology 150, 2569–2576 (2009).
Google Scholar
Coughlan, M. T. et al. RAGE-induced cytosolic ROS promote mitochondrial superoxide generation in diabetes. J. Am. Soc. Nephrol. 20, 742–752 (2009).
Google Scholar
Peppa, M. et al. Fetal or neonatal low-glycotoxin environment prevents autoimmune diabetes in NOD mice. Diabetes 52, 1441–1448 (2003).
Google Scholar
Verdaguer, J. et al. Spontaneous autoimmune diabetes in monoclonal T cell nonobese diabetic mice. J. Exp. Med. 186, 1663–1676 (1997).
Google Scholar
Borg, D. J. et al. Perinatal exposure to high dietary advanced glycation end products in transgenic NOD8.3 mice leads to pancreatic beta cell dysfunction. Islets 10, 10–24 (2018).
Google Scholar
Forbes, J. M. et al. Glucose homeostasis can be differentially modulated by varying individual components of a western diet. J. Nutr. Biochem. 24, 1251–1257 (2013).
Google Scholar
van Dongen, K. C. W. et al. Dietary advanced glycation endproducts (AGEs) increase their concentration in plasma and tissues, result in inflammation and modulate gut microbial composition in mice; evidence for reversibility. Food Res. Int. 147, 110547 (2021).
Google Scholar
Snelson, M. et al. Processed foods drive intestinal barrier permeability and microvascular diseases. Sci. Adv. 7, eabe4841 (2021).
Google Scholar
Beyan, H. et al. Glycotoxin and autoantibodies are additive environmentally determined predictors of type 1 diabetes: a twin and population study. Diabetes 61, 1192–1198 (2012).
Google Scholar
Mericq, V. et al. Maternally transmitted and food-derived glycotoxins: a factor preconditioning the young to diabetes? Diabetes Care 33, 2232–2237 (2010).
Google Scholar
de Courten, B. et al. Diet low in advanced glycation end products increases insulin sensitivity in healthy overweight individuals: a double-blind, randomized, crossover trial. Am. J. Clin. Nutr. 103, 1426–1433 (2016).
Google Scholar
Vlassara, H. et al. Oral AGE restriction ameliorates insulin resistance in obese individuals with the metabolic syndrome: a randomised controlled trial. Diabetologia 59, 2181–2192 (2016).
Google Scholar
Linkens, A. M. et al. A 4-week high-AGE diet does not impair glucose metabolism and vascular function in obese individuals. JCI Insight 7, e156950 (2022).
Google Scholar
Sugaya, K. et al. Three genes in the human MHC class III region near the junction with the class II: gene for receptor of advanced glycosylation end products, PBX2 homeobox gene and a notch homolog, human counterpart of mouse mammary tumor gene int-3. Genomics 23, 408–419 (1994).
Google Scholar
Vissing, H., Aagaard, L., Tommerup, N. & Boel, E. Localization of the human gene for advanced glycosylation end product-specific receptor (AGER) to chromosome 6p21.3. Genomics 24, 606–608 (1994).
Google Scholar
Serveaux-Dancer, M. et al. Pathological implications of receptor for advanced glycation end-product (AGER) gene polymorphism. Dis. Markers 2019, 2067353 (2019).
Google Scholar
Lu, W., Feng, B., Xie, G. & Liu, F. Association of AGER gene G82S polymorphism with the severity of coronary artery disease in Chinese Han population. Clin. Endocrinol. 75, 470–474 (2011).
Google Scholar
Suchankova, P. et al. Is the Gly82Ser polymorphism in the RAGE gene relevant to schizophrenia and the personality trait psychoticism? J. Psychiatry Neurosci. 37, 122–128 (2012).
Google Scholar
Adams, J. N. et al. Genetic analysis of advanced glycation end products in the DHS MIND study. Gene 584, 173–179 (2016).
Google Scholar
Jang, Y. et al. Association of the Gly82Ser polymorphism in the receptor for advanced glycation end products (RAGE) gene with circulating levels of soluble RAGE and inflammatory markers in nondiabetic and nonobese Koreans. Metabolism 56, 199–205 (2007).
Google Scholar
Salonen, K. M. et al. Circulating concentrations of soluble receptor for AGE are associated with age and AGER gene polymorphisms in children with newly diagnosed type 1 diabetes. Diabetes Care 37, 1975–1981 (2014).
Google Scholar
Forbes, J. M. et al. Receptor for advanced glycation end-products (RAGE) provides a link between genetic susceptibility and environmental factors in type 1 diabetes. Diabetologia 54, 1032–1042 (2011).
Google Scholar
Salonen, K. M. et al. A drop in the circulating concentrations of soluble receptor for advanced glycation end products is associated with seroconversion to autoantibody positivity but not with subsequent progression to clinical disease in children en route to type 1 diabetes. Diabetes Metab. Res. Rev. (2016).
Salonen, K. M. et al. Decrease in circulating concentrations of soluble receptors for advanced glycation end products at the time of seroconversion to autoantibody positivity in children with prediabetes. Diabetes Care 38, 665–670 (2015).
Google Scholar
DiMeglio, L. A., Evans-Molina, C. & Oram, R. A. Type 1 diabetes. Lancet 391, 2449–2462 (2018).
Google Scholar
Diabetes Prevention Trial–Type 1 Diabetes Study Group Effects of insulin in relatives of patients with type 1 diabetes mellitus. N. Engl. J. Med. 346, 1685–1691 (2002).
Google Scholar
Skyler, J. S. et al. Effects of oral insulin in relatives of patients with type 1 diabetes: the Diabetes Prevention Trial–Type 1. Diabetes Care 28, 1068–1076 (2005).
Google Scholar
Assfalg, R. et al. Oral insulin immunotherapy in children at risk for type 1 diabetes in a randomised controlled trial. Diabetologia 64, 1079–1092 (2021).
Google Scholar
Krischer, J. P., Schatz, D. A., Bundy, B., Skyler, J. S. & Greenbaum, C. J. Effect of oral insulin on prevention of diabetes in relatives of patients with type 1 diabetes: a randomized clinical trial. JAMA 318, 1891–1902 (2017).
Google Scholar
Ziegler, A. G. et al. Oral insulin therapy for primary prevention of type 1 diabetes in infants with high genetic risk: the GPPAD-POInT (global platform for the prevention of autoimmune diabetes primary oral insulin trial) study protocol. BMJ Open. 9, e028578 (2019).
Google Scholar
Raab, J. et al. Capillary blood islet autoantibody screening for identifying pre-type 1 diabetes in the general population: design and initial results of the Fr1da study. BMJ Open. 6, e011144 (2016).
Google Scholar
Näntö-Salonen, K. et al. Nasal insulin to prevent type 1 diabetes in children with HLA genotypes and autoantibodies conferring increased risk of disease: a double-blind, randomised controlled trial. Lancet 372, 1746–1755 (2008).
Google Scholar
Harrison, L. C. et al. Pancreatic β-cell function and immune responses to insulin after administration of intranasal insulin to humans at risk for type 1 diabetes. Diabetes Care 27, 2348–2355 (2004).
Google Scholar
Ludvigsson, J., Wahlberg, J. & Casas, R. Intralymphatic injection of autoantigen in type 1 diabetes. N. Engl. J. Med. 376, 697–699 (2017).
Google Scholar
Casas, R. et al. Intra-lymphatic administration of GAD-alum in type 1 diabetes: long-term follow-up and effect of a late booster dose (the DIAGNODE extension trial). Acta Diabetol. 59, 687–696 (2022).
Google Scholar
Ludvigsson, J. et al. Intralymphatic glutamic acid decarboxylase with vitamin D supplementation in recent-onset type 1 diabetes: a double-blind, randomized, placebo-controlled phase IIb trial. Diabetes Care 44, 1604–1612 (2021).
Google Scholar
Hannelius, U., Beam, C. A. & Ludvigsson, J. Efficacy of GAD-alum immunotherapy associated with HLA-DR3-DQ2 in recently diagnosed type 1 diabetes. Diabetologia 63, 2177–2181 (2020).
Google Scholar
Ludvigsson, J. et al. Combined etanercept, GAD-alum and vitamin D treatment: an open pilot trial to preserve beta cell function in recent onset type 1 diabetes. Diabetes/Metab. Res. Rev. 37, e3440 (2021).
Google Scholar
Eisenbarth, G. S. et al. Anti-thymocyte globulin and prednisone immunotherapy of recent onset type 1 diabetes mellitus. Diabetes Res. 2, 271–276 (1985).
Google Scholar
Haller, M. J. et al. Anti-thymocyte globulin/G-CSF treatment preserves β cell function in patients with established type 1 diabetes. J. Clin. Investig. 125, 448–455 (2015).
Google Scholar
Haller, M. J. et al. Antithymocyte globulin plus G-CSF combination therapy leads to sustained immunomodulatory and metabolic effects in a subset of responders with established type 1 diabetes. Diabetes 65, 3765–3775 (2016).
Google Scholar
Gitelman, S. E. et al. Antithymocyte globulin treatment for patients with recent-onset type 1 diabetes: 12-month results of a randomised, placebo-controlled, phase 2 trial. Lancet Diabetes Endocrinol. 1, 306–316 (2013).
Google Scholar
Gitelman, S. E. et al. Antithymocyte globulin therapy for patients with recent-onset type 1 diabetes: 2 year results of a randomised trial. Diabetologia 59, 1153–1161 (2016).
Google Scholar
Orban, T. et al. Costimulation modulation with abatacept in patients with recent-onset type 1 diabetes: follow-up 1 year after cessation of treatment. Diabetes Care 37, 1069–1075 (2014).
Google Scholar
Orban, T. et al. Co-stimulation modulation with abatacept in patients with recent-onset type 1 diabetes: a randomised, double-blind, placebo-controlled trial. Lancet 378, 412–419 (2011).
Google Scholar
Russell, W. E. et al. Abatacept for delay of type 1 diabetes progression in stage 1 relatives at risk: a randomized, double-masked, controlled trial. Diabetes Care 46, 1005–1013 (2023).
Google Scholar
Hartemann, A. et al. Low-dose interleukin 2 in patients with type 1 diabetes: a phase 1/2 randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 1, 295–305 (2013).
Google Scholar
Rosenzwajg, M. et al. Low-dose interleukin-2 fosters a dose-dependent regulatory T cell tuned milieu in T1D patients. J. Autoimmun. 58, 48–58 (2015).
Google Scholar
Rosenzwajg, M. et al. Low-dose IL-2 in children with recently diagnosed type 1 diabetes: a phase I/II randomised, double-blind, placebo-controlled, dose-finding study. Diabetologia 63, 1808–1821 (2020).
Google Scholar
Marcovecchio, M. L. et al. Interleukin-2 therapy of autoimmunity in diabetes (ITAD): a phase 2, multicentre, double-blind, randomized, placebo-controlled trial. Wellcome Open. Res. 5, 49 (2020).
Google Scholar
Marwaha, A. K. et al. A phase 1b open-label dose-finding study of ustekinumab in young adults with type 1 diabetes. Immunother. Adv. 2, ltab022 (2022).
Google Scholar
Tatovic, D. et al. Ustekinumab for type 1 diabetes in adolescents: a multicenter, double-blind, randomized phase 2 trial. Nat. Med. 30, 2657–2666 (2024).
Google Scholar
Van Rampelbergh, J. et al. First-in-human, double-blind, randomized phase 1b study of peptide immunotherapy IMCY-0098 in new-onset type 1 diabetes. BMC Med. 21, 190 (2023).
Google Scholar
Van Rampelbergh, J. et al. First-in-human, double-blind, randomized phase 1b study of peptide immunotherapy IMCY-0098 in new-onset type 1 diabetes: an exploratory analysis of immune biomarkers. BMC Med. 22, 259 (2024).
Google Scholar
Pescovitz, M. D. et al. B-lymphocyte depletion with rituximab and β-cell function: two-year results. Diabetes Care 37, 453–459 (2014).
Google Scholar
Pescovitz, M. D. et al. Rituximab, B-lymphocyte depletion, and preservation of beta-cell function. N. Engl. J. Med. 361, 2143–2152 (2009).
Google Scholar
Flandre, T. D., Mansfield, K. G., Espié, P. J., Rubic-Schneider, T. & Ulrich, P. Immunosuppression profile of CFZ533 (Iscalimab), a non-depleting anti-CD40 antibody, and the presence of opportunistic infections in a rhesus monkey toxicology study. Toxicol. Pathol. 50, 712–724 (2022).
Google Scholar
Waibel, M. et al. Investigating the efficacy of baricitinib in new onset type 1 diabetes mellitus (BANDIT) – study protocol for a phase 2, randomized, placebo controlled trial. Trials 23, 433 (2022).
Google Scholar
Waibel, M. et al. Baricitinib and β-cell function in patients with new-onset type 1 diabetes. N. Engl. J. Med. 389, 2140–2150 (2023).
Google Scholar
Louvet, C. et al. Tyrosine kinase inhibitors reverse type 1 diabetes in nonobese diabetic mice. Proc. Natl Acad. Sci. USA 105, 18895–18900 (2008).
Google Scholar
Gitelman, S. E. et al. Imatinib therapy for patients with recent-onset type 1 diabetes: a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Diabetes Endocrinol. 9, 502–514 (2021).
Google Scholar
Ovalle, F. et al. Verapamil and beta cell function in adults with recent-onset type 1 diabetes. Nat. Med. 24, 1108–1112 (2018).
Google Scholar
Forlenza, G. P. et al. Effect of verapamil on pancreatic beta cell function in newly diagnosed pediatric type 1 diabetes: a randomized clinical trial. JAMA 329, 990–999 (2023).
Google Scholar
Wych, J. et al. Investigating the effect of verapamil on preservation of beta-cell function in adults with newly diagnosed type 1 diabetes mellitus (Ver-A-T1D): protocol for a randomised, double-blind, placebo-controlled, parallel-group, multicentre trial. BMJ Open. 14, e091597 (2024).
Google Scholar
von Herrath, M. et al. Anti-interleukin-21 antibody and liraglutide for the preservation of β-cell function in adults with recent-onset type 1 diabetes: a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Diabetes Endocrinol. 9, 212–224 (2021).
Google Scholar
Skyler, J. S. et al. Differentiation of diabetes by pathophysiology, natural history, and prognosis. Diabetes 66, 241–255 (2017).
Google Scholar
Jessop, F. et al. Impairing RAGE signaling promotes survival and limits disease pathogenesis following SARS-CoV-2 infection in mice. JCI Insight 7, e155896 (2022).
Google Scholar
Pickering, R. J. et al. Transactivation of RAGE mediates angiotensin-induced inflammation and atherogenesis. J. Clin. Investig. 129, 406–421 (2019).
Google Scholar
Trivedi, P. M. et al. Repurposed JAK1/JAK2 inhibitor reverses established autoimmune insulitis in NOD mice. Diabetes 66, 1650–1660 (2017).
Google Scholar
Coomans de Brachène, A. et al. IFN-α induces a preferential long-lasting expression of MHC class I in human pancreatic beta cells. Diabetologia 61, 636–640 (2018).
Google Scholar
Ge, T. et al. The JAK1 selective inhibitor ABT 317 blocks signaling through interferon-γ and common γ chain cytokine receptors to reverse autoimmune diabetes in NOD mice. Front. Immunol. 11, 588543 (2020).
Google Scholar
link