Design and setting
This RCT was registered in the Chinese Clinical Trial Registry (ChiCTR2100052301) on 24/10/2021 and approved by the Ethics Committee of the Nanjing Center for Disease Control and Prevention (PJ2021-A001-02). This parallel, investigator-blind, controlled trial was conducted from June 2022 to June 2023 at community health service centers across 12 districts in Nanjing, China. The study adhered to the guidelines of the Helsinki Declaration, and its reporting followed the CONSORT standards. Informed consent was obtained from all participants.
From June to August 2022, a multistage stratified cluster random sampling method was employed to select 1,600 T2DM patients from communities enrolled in the National Essential Public Health Services Program in Nanjing. Eligible patients with mild to moderate chronic periodontitis were recruited via telephone invitations to participate in self-management activities and complete evaluation surveys. The classification criteria for chronic periodontitis and mild to moderate chronic periodontitis were based on previously published studies29,30.
Inclusion/exclusion criteria
Participants were included if they met the following criteria: (1) Aged ≥ 35 years; (2) Permanent residents of Nanjing (residing locally ≥ 6 months in the past year); (3) Diagnosed with T2DM according to the Chinese Guidelines for the Prevention and Treatment of Type 2 Diabetes (2020 edition)2, defined as fasting plasma glucose (FPG) ≥ 7.0 mmol/L, or glycated hemoglobin (HbA1c) ≥ 6.5%, or prior diagnosis by township/higher-level hospitals, or current use of antidiabetic medications/insulin; (4) Enrolled in the National Essential Public Health Services Program for T2DM management; (5) Clinically stable with no severe diabetes complications.
The exclusion criteria included the following: (1) History of infectious diseases (e.g., tuberculosis, syphilis, HIV); (2) Pregnancy; (3) Cognitive impairment or psychiatric disorders; (4) Use of medications affecting glucose metabolism or inducing gingival changes (e.g., immunosuppressants, antiepileptics, calcium channel blockers, steroids).
Postbaseline exclusion process: Of the 1,600 initially screened T2DM patients, 784 were enrolled after excluding those who failed to complete baseline surveys (n = 104), had severe periodontitis (n = 105) or the absence of periodontitis (confirmed via baseline periodontal examination; n = 411) or declined to participate in follow-up interventions (n = 196). The screening process is detailed in Fig. S1.
Sample size
The sample size was estimated using the formula \(N=\frac{2\left({u}_{\alpha }+{u}_{\beta }\right)\times {\sigma }^{2}}{{d}^{2}}\), with a two-sided test α of 0.05, a power (1 − β) of 90%, and an expected HbA1c difference (d) of 0.5% between the intervention and control groups (7.20% vs. 6.70%)31. The estimated standard deviation (σ) was 1.4, and a 10% dropout rate was considered, resulting in a required sample size of 180 participants per group and a total of 720 participants across four groups. Ultimately, 784 T2DM patients with mild to moderate periodontitis were recruited for self-management intervention. The participants were randomly allocated by district, at a 1:1:1:1 ratio to one of four groups: the control group (G0, n = 196), periodontal intervention group (G1, n = 196), diabetes intervention group (G2, n = 196), and comprehensive diabetes and periodontal intervention group (G3, n = 196).
Interventions
For implementation, one community health center was randomly selected in each district to serve as the intervention site for all participants in that district. The three intervention groups formed self-management teams, each consisting of 12–18 T2DM patients with mild to moderate periodontitis. The intervention was based on a 3-month self-management program conducted from September to November 2022, which emphasized the principles of “peer support,” “collaborative participation,” and “self-management.” Each team was co-led by two leaders: (1) a professional leader (e.g., physician, nutritionist) responsible for organizing evidence-based education and chronic disease management, and (2) a patient-elected deputy leader (a peer with T2DM and periodontitis) tasked with facilitating peer interactions, documenting group activities, and addressing members’ health needs. The deputy leader played a critical role in moderating small-group discussions, modeling self-management behaviors (e.g., glucose monitoring, oral hygiene practices), and maintaining peer accountability through regular reminders. Leaders underwent standardized training using the Self-Management Facilitator Manuals for Diabetes and Periodontitis Patients. Training included role-playing, simulated group sessions, and mentorship by the research team. Competency was assessed through trial presentations, and only leaders who passed this assessment were permitted to conduct group activities.
Led by two team leaders, the activities included lectures, group discussions, experience sharing, live demonstrations, and Q&A sessions with community doctors. These activities aimed to equip patients with knowledge, skills, and communication techniques for effective self-management, thereby boosting their confidence. The interventions were held at community health centers or activity rooms, with sessions occurring every two weeks (six sessions total). Each session lasted 1.0–1.5 h and comprised 4–8 subactivities.
The control group (G0) received routine health management according to the National Essential Public Health Services guidelines, with no self-management activities. The intervention group (G1-G3) underwent a specialized self-management model-based intervention in addition to the routine management provided to the control group. The G1 group focused on oral health education, the relationship between periodontal disease and diabetes, periodontal disease treatments (surgical/non-surgical), and regular follow-up maintenance, etc. The detailed activities for the G1 group are listed in Supplementary Table S1. The G2 group focused on diabetes-related knowledge, diabetes self-management skills, psychological adjustment, physical exercise, a healthy diet, medication usage, blood glucose self-monitoring, the management of complications, the maintenance of good lifestyle habits, etc. Detailed activities for the G2 group are listed in Supplementary Table S2. The G3 group received a combined intervention including content from both the G1 and G2 groups, as described in Supplementary Table S3. In addition to the manuals, the intervention utilized tailored educational materials such as PowerPoint presentations and instructional videos. For each intervention group (G1, G2, and G3), specific lesson plans were created, with six different sets of materials aligned with the intervention focus. These videos were created by the project team and regularly updated to ensure accuracy.
Outcome measures and data collection
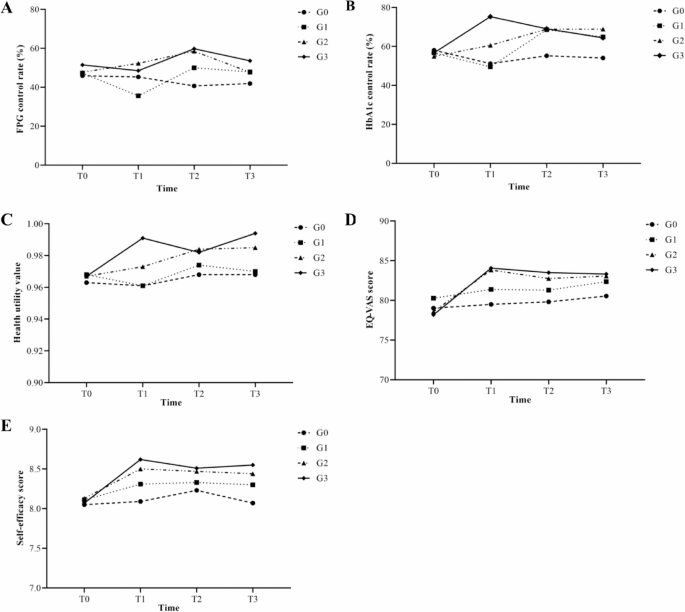
Trained researchers, blinded to group assignments, conducted assessments at baseline (T0), 0 months (immediately postintervention, T1), 3 months (T2), and 6 months (T3) postintervention, during the period from December 1, 2022, to June 1, 2023. Data collection included questionnaires (demographic information, chronic disease self-management, oral health status, etc.), physical measurements (height, weight, etc.), laboratory tests (FPG, HbA1c, etc.), and periodontal examinations. Tablets were used for real-time data collection and validation to ensure accuracy.
Glycemic control
FPG and HbA1c levels were measured according to the Chinese national standard for blood sample collection and processing (GB/T 38,576–2020)32. On the basis of the Chinese Guidelines for the Prevention and Treatment of Type 2 Diabetes (2020 edition), glycemic control status was classified as follows: FPG control status (met: FPG ≤ 7.0 mmol/L; not met: FPG > 7.0 mmol/L) and HbA1c control status (met: HbA1c < 7.0%; not met: HbA1c ≥ 7.0%)2.
Self-efficacy
Self-efficacy was assessed using the eight-item Chronic Disease Self-Management Research Scale, adapted from the Stanford Patient Education Research Center’s scale developed by Lorig et al.33. Each item was rated on a 10-point Likert scale ranging from 1 (not at all confident) to 10 (totally confident). The scale score was calculated as the average of the completed items, with higher scores indicating greater self-efficacy. The scale has demonstrated high reliability and validity in previous studies34.
QoL
QoL was evaluated using the EuroQol Five Dimensions Questionnaire (EQ-5D-5L), which includes five dimensions: mobility, self-care, usual activities (e.g., work, study, housework, family or leisure activities), pain/discomfort, and anxiety/depression). The scores were calculated using the Chinese population-based EQ-5D-5L utility value system, which has demonstrated good reliability and validity35. The health utility value ranges from -0.391 to 1.000, with higher scores indicating better health. An EQ-VAS score was also recorded, where patients self-rated their health on a scale of 0 to 100.
Oral health
Oral health status was assessed on the basis of the methods used in the Fourth National Oral Health Epidemiological Survey Report of China9, covering five items: gingival bleeding (Yes/No), gingival swelling (Yes/No), tooth mobility (Yes/No), chewing weakness (Yes/No), and halitosis (Yes/No).
Healthy lifestyle score
Healthy lifestyle behaviors included: (1) never smoking or having quit smoking; (2) moderate alcohol consumption (< 25 g/day for men, < 15 g/day for women)36; (3) adequate physical activity (≥ 150 min of moderate or ≥ 75 min of vigorous activity per week)37; and (4) a healthy diet, scored on the basis of the frequency of consuming six food groups (vegetables, fruits, whole grains, aquatic products ≥ 1 time/week, soy products ≥ 4 times/week, and red meat < 7 times/week)38. The diet score ranges from 0–6, with a score of 4 or higher considered a healthy diet. Each behavior was scored as 1 (met) or 0 (not met), with a total score ranging from 0 to 4 (higher scores indicating healthier lifestyles)39.
Statistical analysis
Baseline differences between groups were analyzed using ANOVA or the Kruskal‒Wallis H test for continuous variables and the chi-square test for categorical variables. Normality of continuous data was assessed using the Shapiro‒Wilk test. If the data were normally distributed, parametric tests were used (ANOVA); otherwise, non-parametric tests (Kruskal‒Wallis H test) were applied. To assess potential attrition bias, baseline characteristics of completers and dropouts were compared in G0 and G1 using t-tests or Mann‒Whitney U tests for continuous variables and chi-square tests or Fisher’s exact tests for categorical variables. For G2 and G3, descriptive statistics are reported without formal testing due to the small number of dropouts (n ≤ 3). Generalized Estimating Equations (GEEs) were used to analyze longitudinal data, accounting for within-subject correlations, time-varying covariates, and missing data40,41. To address potential intra-district correlations, an unstructured working correlation matrix was utilized within the GEE framework. The GEE model was used to assess the changes over time at different evaluation points for all indicators and to compare the changes in outcome variables between the intervention and control groups. If the group × time interaction was significant (P < 0.05), further analysis was performed to evaluate the main effects of group and time. The model was adjusted for age, gender, area, education level, marital status, occupational status, sleep duration, healthy lifestyle score, duration of T2DM, disease history and medication history. All analyses were performed using IBM SPSS version 23 with a two-sided α of 0.05.
link