To date, most studies on the relationship between dietary patterns and neurodegenerative or psychiatric disorders have focused on a single disease outcome, with limited research integrating multiple outcomes under a unified biological mechanism. In this study, we comprehensively evaluated the association between several healthy dietary patterns and the risk of various neurological and psychiatric conditions, including all-cause dementia, AD, VaD, depression, and anxiety (Supplementary Table 18, Supplementary Fig.12). We observed that higher adherence to healthy dietary patterns was associated with significantly lower risk of neuropsychiatric disorders across multiple models, suggesting a protective effect of diet on brain health. Based on these findings, we explored the mediating roles of systemic inflammation and metabolic biomarkers. The results revealed significant associations between dietary patterns and inflammation levels and suggested that both inflammation and metabolic status partially mediate the relationship between diet and neuropsychiatric outcomes. Although the overall mediating effects were modest, these findings support a potential mechanistic model linking diet, inflammation and metabolism, and brain health.
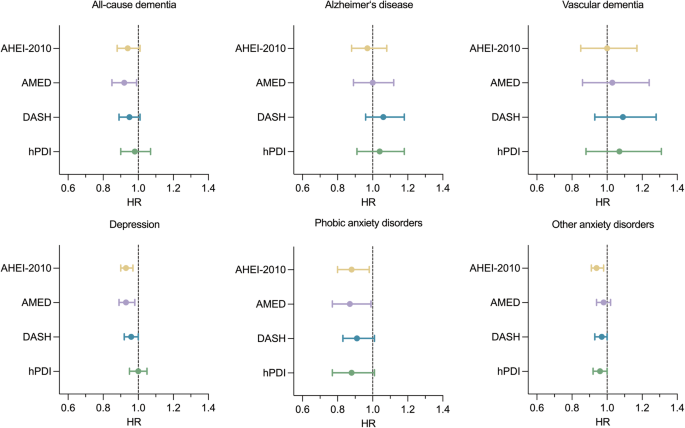
In this study, adherence to the AHEI-2010 was significantly associated with a low risk of depression and anxiety, consistent with previous studies supporting its protective role in mental health21,22. The protective effect of AHEI-2010 appeared stronger for phobic anxiety disorders than for other anxiety disorders, which may be explained by its emphasis on antioxidant-rich foods that reduce oxidative stress and inflammation, potentially modulating amygdala hyperactivation, whereas prefrontal-limbic dysregulation underlying other anxiety disorders may be less responsive to dietary modulation23,24. AMED showed a significant inverse association with all-cause dementia; however, no statistically significant associations were found when examining AD and VaD separately, indicating partial inconsistency with previous findings25,26. These findings may reflect pathophysiological differences. AD is characterized by β-amyloid plaques and tau-related neurofibrillary tangles27, whereas VaD is mainly driven by hypertension- and atherosclerosis-related small vessel disease leading to chronic ischemia, blood-brain barrier dysfunction, and white matter disruption28. Although AMED emphasizes unsaturated fats (including omega-3 fatty acids) and polyphenols with anti-inflammatory and antioxidant properties, these nutrients may have limited impact on AD-specific pathology and are unlikely to sufficiently modify the vascular mechanisms underlying VaD. Additionally, increased adherence to AMED was associated with a low risk of depression29. Notably, this study provides novel evidence suggesting a potential inverse relationship between adherence to AMED and phobic anxiety. Although previous studies have primarily linked the DASH dietary pattern to reductions in depressive symptoms30, its broad role in mental health remains unclear. Our findings extend this evidence by demonstrating associations between adherence to DASH and a low risk of depression and anxiety. In contrast, no significant associations were observed between adherence to hPDI and mental health outcomes.
Notably, the AHEI-2010, DASH, and hPDI scores were not significantly associated with dementia risk in our analysis. Nevertheless, previous studies have shown that high adherence to AMED is associated with large hippocampal volume31 and that hPDI may be linked to reduced risks of dementia and depression32, although some evidence suggests that this protective effect may be limited to males or APOEε4 carriers. Regarding DASH, there is currently a lack of direct evidence supporting its association with reduced dementia risk. Combining the findings of the associations between the four dietary patterns and the risk of dementia, depression, and anxiety, our results support the potential protective effect of overall healthy dietary patterns on mental and psychological health. These dietary patterns share a core foundation of plant-based foods and whole grains, but they differ in specific components and targeted health outcomes. The AHEI-2010 emphasizes balancing various nutrients, with strict scoring for factors such as trans fat restriction, and is most strongly associated with promoting healthy aging as well as maintaining overall physical function and psychological well-being33. The AMED highlights the intake of fish and seafood, legumes, and nuts, while also emphasizing moderate alcohol consumption, which may exert beneficial effects on brain health by attenuating neuroinflammation, improving neurotransmitter metabolism, and promoting healthy brain development34. Despite its cardiovascular origins, the DASH diet conferred modest benefits against depressive symptoms, potentially via vascular-metabolic pathways35, which warrants further investigation in future studies. Collectively, these heterogeneous findings may be explained by the distinct dietary components emphasized in each pattern, differences in scoring algorithms, and the underlying pathophysiological mechanisms of disease subtypes. Additionally, the loss of significance in the associations between DASH and depression and between AMED and all-cause dementia in sensitivity analyses may partly reflect reverse causality. Moreover, the protective effects of dietary patterns may require longer follow-up to become evident, consistent with the cumulative nature of dietary influences on mental and cognitive health. In particular, the attenuation after excluding early cases suggests that baseline dietary characteristics may have been influenced by subclinical disease in individuals who developed outcomes within the first 2 years, and that the protective effects of DASH are therefore more relevant to primary prevention rather than secondary prevention contexts.
The AMED, AHEI-2010, DASH, and hPDI dietary patterns emphasize a high intake of plant-based foods, including vegetables, fruits, whole grains, legumes, seeds, and nuts. These foods are rich in anti-inflammatory and antioxidant compounds, such as flavonoids, polyphenols, and vitamins C and E. Long-term flavonoid intake has been associated with a low risk of AD and related dementia, primarily through improvements in memory, attention, and executive functioning36. Additionally, flavonoid consumption has been linked to reduced depressive symptoms and dementia risk, with the strongest protective effects observed among individuals with high genetic risk, hypertension, or preexisting depressive symptoms37. Seeds and nuts are rich in omega-3 fatty acids, especially alpha-linolenic acid, which has strong anti-inflammatory properties. These fatty acids may contribute to neuroprotection by suppressing proinflammatory cytokine production and modulating neuroinflammatory processes. Oxidative stress plays a pivotal role in the pathogenesis and mechanisms of dementia, which causes neuronal damage38,39,40. Given that oxidative stress is a key driver of inflammation and is strongly implicated in neuronal damage, brain aging, neurodegenerative diseases, and mood disorders, dietary antioxidants—such as vitamins C and E and polyphenols—may play a vital role in preserving cognitive function and preventing neurodegeneration41. Additionally, different dietary patterns have been shown to shape distinct gut microbiota compositions. Healthy dietary interventions, such as the Mediterranean diet, can enrich the gut microbiota and, through modulation of the microbiota–gut–brain axis, contribute to improvements in cognition and emotional regulation42.
However, other essential neuroprotective nutrients—such as vitamins B and D—are predominantly found in animal-sourced foods, including lean meat, eggs, dairy products, fish, and seafood. High intake of vitamin B has been inversely associated with stress-related symptoms and may protect against depression and anxiety43. Similarly, vitamin D, which is involved in cell growth, neuromuscular function, and immune regulation, has been associated with a low risk of major depressive disorders and anxiety44,45. Most clinical trials have suggested that vitamin D supplementation alleviates symptoms of depression and anxiety46. Moreover, minerals and trace elements are essential for the survival and growth of some gut bacteria, while deficiencies or toxicities promote the enrichment of pathogenic bacteria that trigger gut inflammation42.Therefore, the limited intake of animal-derived foods in plant-based diets, particularly strict hPDI, may contribute to the lack of significant associations with reduced risks of dementia, depression, and anxiety observed in this study. However, when analyzing the correlations of hPDI with inflammatory and metabolic markers, hPDI was inversely associated with most inflammatory indicators and showed significant correlations with the lipid composition of VLDL, but these potential benefits may have been offset. Additionally, dietary ω-3 PUFAs from fish consumption are associated with higher cognitive function and exert protective effects against depression47,48. These findings highlight the potential limitations of strict plant-based dietary patterns for providing sufficient neuroprotective nutrients. However, vegetable oils, which were not available in the UKB database, may have anti-inflammatory properties, and this requires further investigation.
Moreover, several studies have shown that the consumption of red and processed meat is associated with altered serum lipid levels, such as triglycerides, and positively correlated with higher serum CRP levels49,50,51. Furthermore, ultra-processed foods are recognized as triggers of low-grade systemic inflammatory and oxidative changes52,53. Chronic low-grade inflammation is a well-established contributor to the development of cardiovascular diseases, diabetes, neurodegenerative disorders, and mood-related conditions54,55,56. Consequently, excessive and long-term consumption of processed meat may increase the risk of these health outcomes. Similarly, frequent consumption of sweets, including cakes, candies, and sugar-sweetened beverages, can result in rapid spikes in blood glucose levels, increased insulin secretion, and fat accumulation, which may trigger oxidative stress and systemic inflammation57. This may partially explain their detrimental associations with cognitive and mental health outcomes.
These mechanistic insights are largely consistent with the associations observed between healthy dietary pattern adherence and a reduced risk of neurocognitive and mood disorders. In this study, which was grounded in a unified biological framework, we systematically examined the interrelationships among diet, inflammation, and common neuropsychiatric disorders. Our findings support the hypothesis that dietary patterns influence systemic inflammation primarily by modulating metabolic function. Several healthy dietary patterns were significantly associated with low levels of multiple circulating inflammatory markers, indicating their potential anti-inflammatory effects. In the mediation analysis, we initially incorporated multiple inflammatory indicators, including CRP, INFLA-score, neutrophil-to-lymphocyte (NLR) and platelet-to-lymphocyte (PLR), but the overall model fit was poor, and only CRP exhibited a consistent and statistically significant mediating effect. This may reflect the sensitivity of CRP in detecting chronic systemic inflammation, which has been implicated in triggering or exacerbating neuroinflammatory processes. The observed inverse association between dietary patterns and CRP levels may be partly explained by the high intake of plant-based foods, such as vegetables and fruits, which are rich in anti-inflammatory and antioxidant compounds.
Additionally, we observed that long-term adherence to healthy dietary patterns may influence neuropsychiatric outcomes by improving metabolic function. Previous prospective studies have consistently identified metabolic dysregulation, such as impaired glucose metabolism and dyslipidemia, as the major risk factor for dementia, depression, and anxiety58,59,60. Our results suggest that diet-induced metabolic changes may contribute to disease prevention. Notably, we found a strong correlation between metabolic and inflammatory profiles, indicating that diet-driven alterations in small-molecule metabolites may influence inflammatory signaling pathways, thereby exerting either proinflammatory or anti-inflammatory effects and ultimately affecting the risk of neuropsychiatric disorders. In our analysis, several metabolites and pathways appeared to play a central role in this process, including the composition of lipids in very large VLDL particles — specifically, the ratios of cholesterol and cholesteryl esters to total lipids and triglycerides to total lipids — and the overall proportion of polyunsaturated fatty acids (PUFAs). The lipid composition of very large VLDL particles has been linked to insulin resistance, type 2 diabetes, and coronary artery calcification, all of which are known to contribute to the development of cognitive impairment and mental disorders61. Among the PUFAs, omega 3 polyunsaturated fatty acids (n-3 PUFAs) obtained from the diet are essential. They exert anti-inflammatory effects and modulate lipid metabolism by inhibiting inflammatory signaling pathways, such as nuclear factor-κB (NF-κB), down-regulating fatty acid synthesis via sterol regulatory element binding protein-1c (SREBP-1c), and up-regulating fatty acid oxidation through peroxisome proliferator-activated receptor α (PPARα). In addition, the incorporation of n-3 PUFAs into cell membrane phospholipids enhances membrane fluidity, which can lead to modifications in the way transmembrane proteins, such as receptors, interact with their ligands (Supplementary Tables 19–21, Supplementary Figure 13)62,63.
The biological pathways explored in this study provide additional evidence of the protective role of diet in common age-related neuropsychiatric conditions. Our findings deepen our current understanding of the diet-metabolism-inflammation-disease axis and offer a theoretical foundation for preventive strategies based on nutritional interventions. Given that dietary patterns are modifiable and sustainable lifestyle factors, beneficial patterns (such as AMED, AHEI-2010, and DASH) may inform personalized dietary recommendations, the development of public health nutrition guidelines, and multidisciplinary intervention strategies targeting inflammation and metabolic regulation. These strategies include population-level dietary guidelines, community-based education programs, and personalized nutrition approaches that consider genetic and lifestyle risk factors. Moreover, at the clinical level, integrating dietary counseling into routine care may further facilitate early prevention among high-risk individuals. Furthermore, this study highlights the importance of specific neuroprotective nutrients in maintaining cognitive and mental health and offers potential directions for early screening and supplementation strategies in older populations.
This study has some limitations. First, dietary patterns are not static and may change over time owing to factors such as demographic location, environmental influences, lifestyle modifications, or major health events. Meanwhile, although the cultural adaptability of the AMED score has been internationally recognized, the inclusion of alcohol consumption may not be appropriate in non-Mediterranean regions, highlighting the need for future studies to explore localized dietary scoring systems that better reflect cultural dietary practices. Second, both diet-induced metabolic alterations and chronic inflammation are cumulative processes that typically evolve over long periods. Therefore, long-term follow-up may be needed to fully capture their long-term effects. Additionally, dietary pattern scores were derived using tertile-based categorization. While this approach facilitates group comparisons, it may not fully reflect actual dietary intake levels, potentially underestimating inter-individual variability and limiting the assessment of potential dose-response relationships. Although our study included a broad range of metabolomic and inflammatory biomarkers across multiple pathways, these markers may not capture the complexity of the body’s metabolism-inflammatory network. Finally, as the study population primarily consisted of middle-aged and older adults, the generalizability of the findings to younger or more diverse populations warrants further investigation.
In conclusion, adherence to healthy dietary patterns—particularly AMED and AHEI-2010—was associated with lower risks of dementia, depression, and anxiety. The associations between dietary patterns and neuropsychiatric outcomes are likely mediated through inflammatory and metabolic pathways, highlighting the importance of precision-targeted dietary recommendations that modulate the diet-immune-brain axis. Our findings suggest that adherence to healthy dietary patterns could represent a promising public health strategy for the prevention of neuropsychiatric disorders. However, further research, including interventional studies, is needed to confirm causal relationships and clarify the underlying mechanisms.
link