Data source
The national “Happy Breathing” Programme is a prospective, nationwide, ongoing cohort study that was initiated on November 3, 2018, in China. The programme, led by the Respiratory Disease Medical Consortium and the National Clinical Research Center for Respiratory Diseases [16], aims to enroll COPD patients aged 40 years and older and enhance the effectiveness of COPD care. To ensure comprehensive coverage, 32 administrative regions were randomly selected following stratification by economic rank, making the sample theoretically nationally representative [17]. However, despite the initial random selection approach, some regions that were approached declined to participate in the “Happy Breathing” Programme. This may have introduced some selection bias and limited the representativeness of the final sample. Nevertheless, this remains the largest COPD cohort in China to date.
Patients were identified and recruited both actively and passively; they accessed the healthcare system during the study period [16]. For active case finding, physicians at participating health care facilities screened their patients using the COPD Screening Questionnaire (COPD-SQ) as part of the “Happy Breathing” Programme. Those with a COPD-SQ score above 16 and who provided consent underwent post-bronchodilator pulmonary function testing (PFT) [18]. Patients with an FEV1/FVC ratio below 0.7 on the PFT were diagnosed with COPD based on Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines [19]. For passive case finding, patients with a pre-existing, clinically confirmed COPD diagnosis were also enrolled, provided they gave informed consent (Fig. 1). Additionally, patients with a physician diagnosis of asthma or asthma–COPD overlap syndrome were excluded during the enrollment process, ensuring that the cohort reflected the individuals with a clinical diagnosis of COPD only. More details on the inclusion and exclusion criteria for COPD patients can be found in the study protocol [17].
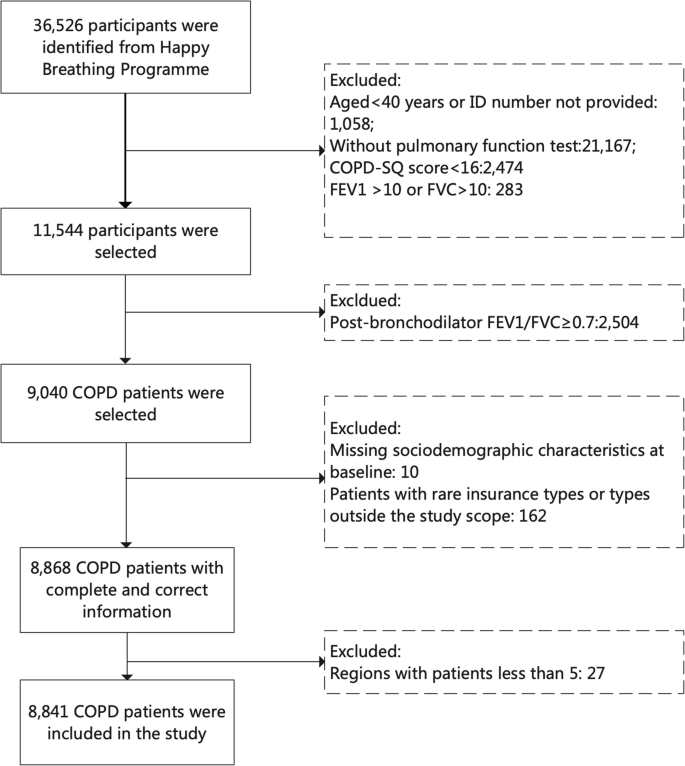
Flow diagram of sample selection
Overall, the study utilized baseline data from the “Happy Breathing” Programme collected between November 2018 and April 2024, and our sample represents individuals who sought care within the Chinese healthcare system. Findings based on this sample can offer valuable insights into the reform and optimization of insurance policies to improve the COPD care cascade in China.
COPD care cascade outcome measures
The concept of the care cascade has gained prominence in public health studies as a crucial tool for evaluating disease management. It helps identify gaps in healthcare delivery and guides efforts to address them [15]. Additionally, it can serve as a benchmark for setting aspirational goals, enabling health systems to enhance care for the populations most in need [20, 21]. We defined the COPD care cascade in five stages: (1) tested, (2) diagnosed, (3) treated, (4) controlled with mild or no exacerbations in the past year (controlled 1), and (5) controlled with no exacerbations in the past year (controlled 2) [16].
Patients were defined as tested if they answered “yes” to either of the following questions: “Have you received a PFT?” or “Have you ever received a PFT in the past?”. Patients were diagnosed if they were tested and answered “yes” to the question: “Have you ever been diagnosed with COPD?”. Patients were considered treated if they had been diagnosed and reported receiving inhaled medications recommended by COPD treatment guidelines [22]. Patients were classified as controlled 1 if they were diagnosed with COPD and answered “no” to the question: “Have you been hospitalized in the past 12 months due to acute exacerbation?”. Similarly, patients were classified as controlled 2 if they had no COPD exacerbations in the past 12 months and answered “no” to the question: “Have you experienced any acute exacerbations in the past 12 months?” [23].
Basic medical insurance coverage and covariates
In China, basic medical insurance is a government-established system designed to reduce the financial burden of illness for its residents. By the time baseline data for the “Happy Breathing” Programme were collected, most regions had two main insurance schemes in operation: UEBMI and URRBMI [24]. The reimbursement rates for medical expenses under UEBMI and URRBMI are different, with URRBMI enrollees generally facing higher deductibles and copayments than UEBMI enrollees [25, 26]. These rates may vary depending on the region, specific policies, whether the service is outpatient or inpatient, and the level of the healthcare institution [27, 28]. More details on the different types of basic medical insurance and their reimbursement policies for COPD in China are provided in Additional file 1: Text S1 [29,30,31,32,33,34,35,36].
Patients’ insurance coverage status was determined based on their self-reported response to the survey question: “What is your payment method for medical treatment?” The response options included various basic medical insurance types, commercial health insurance, full public funding, uninsured, and other self-specified insurance. We classified each patient’s insurance coverage status as a categorical variable with six distinct categories based on their responses: (1) URRBMI, (2) UEBMI, (3) commercial health insurance, (4) fully covered by public funds, (5) uninsured, and (6) other insurance.
Covariates were selected based on Andersen’s Healthcare Utilization Model and derived from baseline survey data and physical measurements [37]. They were adjusted for in the evaluation of the association between health insurance and cascade outcomes. Andersen’s Healthcare Utilization Model provides a theoretical framework for selecting individual-level variables in our analysis. It conceptualizes healthcare utilization as being determined by three core components. The first component is “predisposing factors.” Accordingly, we included age, gender, education, occupation, place of residence (rural/urban), body mass index (BMI), and smoking status as covariates. Age was categorized as < 65 years and ≥ 65 years. Education level was classified as “below high school” and “high school or above.” Occupation was categorized as “unemployed” and “employed.” BMI was grouped into underweight (< 18.5 kg/m2), healthy weight (18.5–23.9 kg/m2), and overweight or obese (≥ 24.0 kg/m2). Smoking status was classified as “non-smoker,” “ex-smoker,” and “current smoker” based on participants’ responses to the survey question on smoking behavior. Those who reported “never smoked” were categorized as non-smokers; those who selected “formerly smoked (and have quit for ≥ 6 months)” were classified as ex-smokers; and those who indicated “currently smoking or have quit for less than 6 months” were categorized as current smokers. The second component is “enabling factors,” represented in our analysis by participants’ insurance status. The third component is “need factors,” which refer to individuals’ perceived need for care. As we used baseline data from the “Happy Breathing” Programme, which is collected before any programme-related interventions, we assumed that patients’ perceived need was not yet influenced by the programme.
The missing data for the variables mentioned above accounted for approximately 1.9% of the total sample, with 1.2% due to missing values in insurance status and the remainder primarily due to missing data on occupation and education. These low levels of missingness in key socioeconomic and insurance variables suggest that our study sample is unlikely to be substantially biased due to missing data.
Statistical analysis
We calculated the proportion of individuals reaching each care cascade step (tested, diagnosed, treated, controlled 1, and controlled 2) among the participants stratified by self-reported insurance coverage status. To evaluate the association between medical insurance coverage and the likelihood of reaching each care cascade step, we applied generalized linear regression models with a Poisson distribution, the log link function, and robust standard errors, i.e., modified Poisson regression for binary outcomes [38]. The estimated relative risk (RR) provided a consistent estimate of the average effect and was deemed easier to interpret than an odds ratio estimated from logistic regression [38,39,40,41,42]. As insurance policies are regulated and under supervision at the regional level, we applied region fixed effects to control for variations in insurance reimbursement policies across geographic regions. Standard errors were also adjusted for clustering at the regional level. A p-value < 0.05 was considered statistically significant in all analyses.
Additionally, we conducted stratified analyses by age group, sex, occupation, and residential location to examine whether the association between basic medical insurance schemes and COPD management varies across major demographic subgroups.
Lastly, we conducted a range of sensitivity analyses to check the robustness and consistency of our findings under different model specifications and assumptions. China’s reimbursement system for COPD is highly complex and varies significantly across regions (Additional file 1: Text S1). At the time of data collection, both diagnosis and testing for COPD were covered under basic medical insurance schemes. However, while some regions had gradually included COPD treatment in their outpatient reimbursement lists (ORL), others provided little or no coverage for outpatient COPD treatment [43]. Given the continuous enrollment in the “Happy Breathing” Programme, patients’ eligibility for outpatient reimbursement depended on whether their prescriptions were issued after COPD had been included in their region’s ORL. To address this variability, we first conducted a sensitivity analysis comparing outcomes for patients in regions where COPD had been added to the ORL (defined by region-year) with those in regions where COPD had not yet been included (see Additional file 1: Table S1 for a complete list of regions and the dates they included COPD in their ORL).
According to the 2024 GOLD guidelines [44], the treatment of COPD includes not only inhaled therapies but also non-inhaled pharmacological treatments and non-pharmacological interventions. Therefore, in our second sensitivity analysis, we broadened the definition of COPD treatment to encompass a wider range of therapeutic modalities. This allowed us to further assess the association between basic medical insurance coverage and this broader definition of treatment, as well as its relationship with subsequent exacerbation control. In this analysis, non-inhaled pharmacological treatments included oral β-agonists (e.g., salbutamol, terbutaline), intravenous or oral theophyllines, expectorants, systemic corticosteroids, and traditional Chinese medicine or proprietary Chinese medicines. Non-pharmacological interventions included influenza vaccination, pneumococcal vaccination, smoking cessation counseling, long-term oxygen therapy, and home non-invasive positive pressure ventilation (NIPPV). Notably, several of these non-pharmacological interventions, such as smoking cessation counseling, long-term oxygen therapy, home NIPPV, and vaccinations, are not currently covered under the reimbursement scope of basic medical insurance. This lack of coverage may limit both the accessibility and adherence to these treatments among patients.
In the third sensitivity analysis, women in China often have a low prevalence of smoking but higher exposure to household air pollution due to the use of biomass fuels for cooking. To assess the potential confounding effect of biomass fuel exposure on the association between medical insurance and COPD management, we extracted data from the survey question: “Do you primarily use biomass fuel (e.g., corn stalks, corn cobs, coal, animal dung) for cooking or heating?”, with response options of “yes” or “no”. We included a binary indicator of biomass fuel exposure as a covariate in the modified Poisson regression model to examine whether the observed associations remained robust after adjustment.
All data and statistical analyses were performed in R (version 4.1.1), and the fixed effects models were implemented using the fixest package version 0.10.1 in R.
Ethics and dissemination
Ethical approval was obtained from the China–Japan Friendship Hospital (approval number: 2019–41-k29). All participants provided written informed consent for participation. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki.
link