Data source and study population
We examined the NHIS–NSC from the Korean NHIS, which covers all residents living in Korea37,38,39. The NHIS-NSC is consisted of a sample of 1 million Korean individuals who were enrolled in health insurance and received medical benefits in Korea. The NHIS–NSC represents approximately 2.2% of the general Korean population and covers the period from 2002 to 2019.
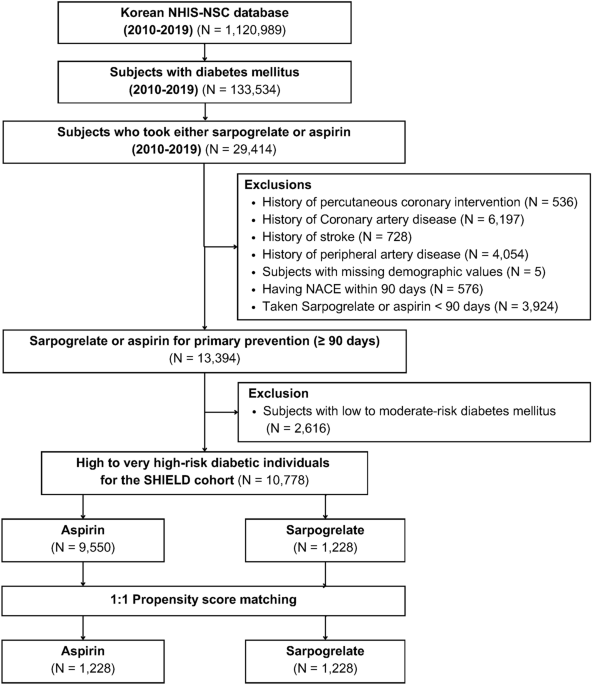
We targeted individuals with DM who were receiving either sarpogrelate or aspirin as single-antiplatelet therapy between 2010 and 2019. Individuals with DM were defined as those who met at least one of the following criteria: use of DM medications, or fasting blood glucose levels of 126 mg/dL or higher40,41,42.
To ensure the study targeted primary prevention settings, we excluded those with a history of coronary artery disease ICD-10 codes: I20-I22), procedure codes for those who had undergone percutaneous coronary intervention, stroke (hospitalization due to ICD-10 code: I63-I64 and computed tomography or magnetic resonance imaging of the brain performed within 90 days before and after admission] or PAD (ICD-10 code: I73.9) were excluded from the study. Additionally, subjects with missing demographic data were excluded.
The study was approved by the Institutional Review Board of Gachon University Gil Medical Center, Incheon, Korea (GFIRB2023-459). Written informed consent was waived by the Institutional Review Board of Gachon University Gil Medical Center, Incheon, Korea, as this is a retrospective study of de-identified administrative data. All methods were performed in accordance with the relevant guidelines and regulations.
Definition and risk assessment of diabetes
Among the selected subjects, only those classified as high-risk or very-high-risk for diabetes were included7,8. The high-risk group was defined as individuals diagnosed with DM for 10 years or more and having at least one of the following conditions: age ≥ 50, hypertension, dyslipidemia (ICD-10 code E78, taking statins or low-density lipoprotein (LDL) cholesterol ≥ 130 mg/dL), current smoker, or BMI ≥ 25. The very-high-risk group was defined as individuals with target organ damage or having at least three of the following conditions: age ≥ 50, hypertension, dyslipidemia, current smoker, or BMI ≥ 257,8. Target organ damage was defined as having diabetic retinopathy (ICD-10 codes H36.0, E10.3, E11.3, E12.3, E13.3, or E14.3)43, diabetic neuropathy (a diagnosis of DN [ICD-10 codes E10.4, E11.4, E12.4, E13.4, E14.4, G59.0, G63.2, and G99.0] or receipt of a prescription for glucose-lowering drugs [ATC code A10B or A10A] plus drugs for diabetic neuropathy [ATC codes A16AX01, D11AX02, N06AA10, N06AA09, N03AX12, N03AX16, and N06AX21])44, or diabetic nephropathy (ICD-10 codes ICD-10 codes E10.2, E11.2, E12.2, E13.2, E14.2, or N08.3)45. The use of insulin at baseline was defined as insulin administration for more than 3 months within the year prior to the initiation of aspirin or sarpogrelate.
Aspirin and sarpogrelate therapy
We selected patients who had been on aspirin (100 mg daily) or sarpogrelate (either 100 mg twice daily or 300 mg once daily) single-antiplatelet therapy for at least 90 days. To ensure all participants were consistently on therapy throughout follow-up, we collected records of the dates and durations of drug prescriptions. We considered full adherence when the total number of prescribed days was equal to or greater than the follow-up duration. The adherence rate during follow-up was determined by calculating the proportion of total prescribed days relative to the total number of follow-up days.
Definition of other comorbidities and laboratory data
Hypertension was defined by diagnosis (I10), anti-hypertensive medication administration, or blood pressure ≥ 140/90 mmHg recorded during health checkup46,47. Data on blood pressure, fasting blood glucose, BMI, LDL cholesterol, high-density lipoprotein cholesterol, triglyceride, creatinine, and hemoglobin were obtained from the national health examination conducted within the two years prior to the specific drug initiation. LDL cholesterol was calculated based on the Friedewald Eq. 48,49,50 For subjects with missing values from the national health examination data, mean values according to each DM risk group were imputed. Statin therapy, including statin-ezetimibe single-pill combination therapy, was defined as its use for more than 3 months prior to the initiation of the antiplatelet.
Clinical follow-up and definition of endpoints
The primary endpoint was NACE, defined as a composite of efficacy and overall bleeding. The efficacy endpoint was a composite of all-cause death, MI, and stroke. Overall bleeding was defined as composite of ICH, and GI bleeding. Other individual secondary endpoints were all-cause death, MI, stroke, GI bleeding, and ICH. These events were identified using the ICD-10 and Medical Care Procedure codes: MI (I21), stroke (hospitalization with I63-I64 and CT/MRI performed within a 90-day window of hospitalization), ICH (hospitalization with I60-I62 or clinic visit for I60-I62 and transfusion), and GI bleeding. GI bleeding was defined as endoscopic hemostasis, vascular embolization combined with specific ICD-10 codes or transfusion accompanied by specific ICD-10 codes and endoscopic examination (E7611, EZ937, E7660, E7670, E7680) within 30 days before or after the transfusion. The ICD-10 codes for GI bleeding are K26.0, K26.2, K26.4, K26.6, K27.0, K27.2, K27.4, K27.6, K28.0, K28.2, K28.4, K28.6, K29.0, K62.0, K92.1, and K92.2. Both groups were followed until the onset of the first NACE event, or the end of study (December 31, 2019). Individuals who experienced a NACE event within 90 days of the initial drug prescription were excluded to minimize reverse causality. Those who took sarpogrelate or aspirin for less than 30 days were also excluded.
Hospitalization due to bleeding was defined as ICH or GI bleeding events that occurred during hospitalization. Fatal bleeding was defined as cases where ICH or GI bleeding was documented among patients who died during hospitalization. Deaths due to bleeding that occurred outside of a hospital setting were excluded from this analysis because they could not be accurately captured using Korean NHIS.
Statistical analysis
PSM was performed in a 1:1 ratio (caliper width: 0.2) to compare the effect on NACE51,52,53. In the pre-PSM cohort, key differences between the two groups were included as covariates. Covariates for matching included sex, age, hypertension, diabetic retinopathy, diabetic neuropathy, diabetic nephropathy, atrial fibrillation, heart failure, statin therapy, fasting blood glucose, BMI, and duration of DM.
Chi-square tests, t-tests, and ANOVA were performed to compare the baseline characteristics based on the intake of antiplatelet therapy. A Cox proportional hazard regression model was used to estimate the HR and 95% CI while adjusting for covariates. The survival rates of NACE were calculated using the Kaplan–Meier method by the intake of specific drugs. Statistical significance was set at p < 0.05. All statistical analyses were performed using SAS Enterprise version 8.3 (SAS Institute, Cary, NC, USA) and R version 4.3.0 (R studio, PBC).
link