National Library of Medicine (NLM). MeSH Descriptor Data 2021: Hearing Loss. (2021).
Luxon, L. M. Hearing Loss. In Encyclopedia of the Neurological Sciences 2nd edn (eds. Aminoff, M. J. & Daroff, R. B.) 533–538 (Academic Press, Oxford, 2014).
World Health Organization. Deafness and hearing loss. Geneva: World Health Organization. (2025).
World Health Organization. World report on hearing. Geneva: World Heath Organization. (2021).
GBD 2019 Hearing Loss Collaborators. Hearing loss prevalence and years lived with disability, 1990–2019: findings from the Global Burden of Disease Study 2019. Lancet 397, 996–1009 (2021).
Google Scholar
Olusanya, B. O. et al. Developmental disabilities among children younger than 5 years in 195 countries and territories, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Glob. Health 6, e1100–e1121 (2018).
Google Scholar
Brown, C. S., Emmett, S. D., Robler, S. K. & Tucci, D. L. Global hearing loss prevention. Otolaryngol. Clin. North Am. 51, 575–592 (2018).
Google Scholar
Kraaijenga, V. J. C. et al. Cochlear implant performance in children deafened by congenital cytomegalovirus—a systematic review. Clin. Otolaryngol. 43, 1283–1295 (2018).
Google Scholar
Hilditch, C. et al. Does screening for congenital cytomegalovirus at birth improve longer term hearing outcomes? Arch. Dis. Child. 103, 988 (2018).
Google Scholar
Deltenre, P. & Van Maldergem, L. Chapter 158—Hearing loss and deafness in the pediatric population: causes, diagnosis, and rehabilitation. in Handbook of Clinical Neurology, Vol. 113 (eds Dulac, O., Lassonde, M. & Sarnat, H. B.) 1527–1538 (Elsevier, 2013).
Tarshish, Y., Leschinski, A. & Kenna, M. Pediatric sudden sensorineural hearing loss: diagnosed causes and response to intervention. Int. J. Pediatr. Otorhinolaryngol. 77, 553–559 (2013).
Google Scholar
Wilson, B. S., Tucci, D. L., O’Donoghue, G. M., Merson, M. H. & Frankish, H. A Lancet Commission to address the global burden of hearing loss. Lancet 393, 2106–2108 (2019).
Google Scholar
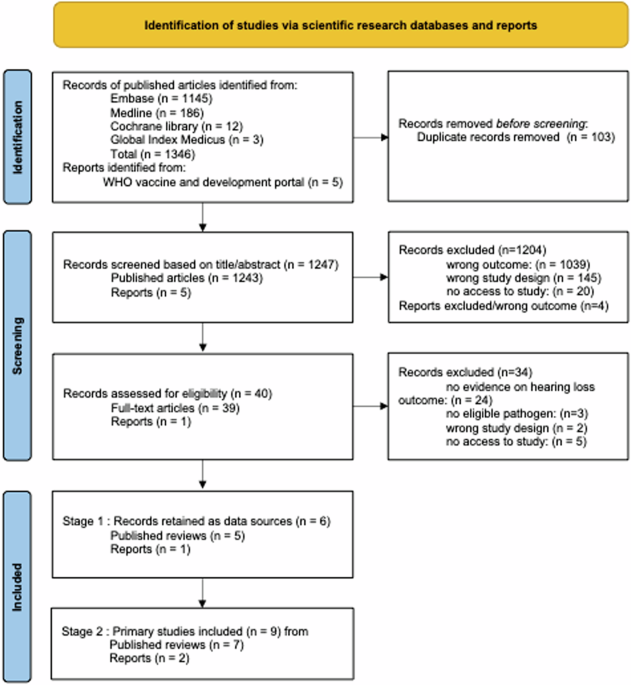
Peters, M. D. J. et al. Chapter 11: Scoping reviews (2020 version). in JBI Manual for Evidence Synthesis (eds Aromataris, E. & Munn, Z.) (JBI, 2020).
Tricco, A. C. et al. PRISMA extension for scoping reviews (PRISMA-ScR): checklist and explanation. Ann. Intern. Med. 169, 467–473 (2018).
Google Scholar
Johri M. & Wilson, B. S. LCOHL: Vaccination for prevention of worldwide hearing loss. Vol. 2023 (OSF, 2023).
World Health Organization. Immunization, Vaccines and Biologicals: Vaccine-Preventable Diseases (including pipeline vaccines). (2025).
World Health Organization. Strategic Advisory Group of Experts on Immunization (SAGE): WHO Vaccine Position Papers. (2025).
World Health Organization. Table 1: Summary of WHO Position Papers – Recommendations for Routine Immunization. (2024).
European Centre for Disease Prevention and Control. Vaccination schedules for individual European countries and specific age groups. Available from: (2024).
Centers for Disease Control and Prevention. Child and Adolescent Immunization Schedule by Age : Recommendations for Ages 18 Years or Younger. (2025).
Centers for Disease Control and Prevention. Adult Immunization Schedule by Age Recommendations for Ages 19 Years or Older. (2025).
Government of Canada. Table 1: Routine childhood immunization schedule, infants and children (birth to 17 years of age). (2024).
Government of Canada. Vaccination for adults. (2023).
World Health Organization. WHO Vaccine Pipeline Tracker. (2025).
Serajuddin, U. & Hamadeh, N. New World Bank country classifications by income level: 2020-2021. in World Bank Blogs (Data Blog, 2020).
Grimshaw, J. A Knowledge Synthesis Chapter. in A Guide to Knowledge Synthesis (Canadian Institutes of Health Research, 2010).
World Health Organization. Product & Delivery Research. (2025).
World Health Organization. Product and delivery research. (2025).
World Health Organization. Handbook on health inequality monitoring: with a special focus on low- and middle-income countries. (2013).
van Heerbeek, N. et al. Effect of combined pneumococcal conjugate and polysaccharide vaccination on recurrent otitis media with effusion. Pediatrics 117, 603 (2006).
Google Scholar
Straetemans, M., Palmu, A., Auranen, K., Zielhuis, G. A. & Kilpi, T. The effect of a pneumococcal conjugate vaccine on the risk of otitis media with effusion at 7 and 24 months of age. Int. J. Pediatr. Otorhinolaryngol. 67, 1235–1242 (2003).
Google Scholar
Menser, M. A., Hudson, J. R., Alan, M. M. & Laurence, J. U. Epidemiology of congenital rubella and results of rubella vaccination in Australia. Rev. Infect. Dis. 7, S37–S41 (1985).
Google Scholar
Le, T.-M., Rovers, M. M., Veenhoven, R. H., Sanders, E. A. M. & Schilder, A. G. M. Effect of pneumococcal vaccination on otitis media with effusion in children older than 1 year. Eur. J. Pediatr. 166, 1049–1052 (2007).
Google Scholar
Hashimoto, H., Fujioka, M., Kinumaki, H. & Kinki Ambulatory Pediatrics Study Group. An office-based prospective study of deafness in mumps. Pediatr. Infect. Dis. J. 28, 173–175 (2009).
Cheffins, T., Chan, A., Keane, R. J., Haan, E. A. & Hall, R. The impact of rubella immunisation on the incidence of rubella, congenital rubella syndrome and rubella-related terminations of pregnancy in South Australia. BJOG Int. J. Obstet. Gynaecol. 105, 998–1004 (1998).
Google Scholar
Darin, N. & Hanner, P. Changes in prevalence, aetiology, age at detection, and associated disabilities in preschool children with hearing impairment born in Göteborg. Dev. Med. Child Neurol. 39, 797–802 (1997).
Google Scholar
Dayan, G. H. et al. Recent resurgence of mumps in the United States. N. Engl. J. Med. 358, 1580–1589 (2008).
Google Scholar
Gao, Z. et al. Models of strategies for control of rubella and congenital rubella syndrome—a 40 year experience from Australia. Vaccine 31, 691–697 (2013).
Google Scholar
World Health Organization. WHO recommends R21/Matrix-M vaccine for malaria prevention in updated advice on immunization. (World Health Organization, 2023).
U.S. Food & Drug Administration. Respiratory Syncytial Virus (RSV). (2024).
World Health Organization. A New Vaccine Will Change the Balance of the Fight Against Meningitis (World Health Organization, 2023).
Kenna, M. A. Acquired hearing loss in children. Otolaryngol. Clin. North Am. 48, 933–953 (2015).
Google Scholar
Cohen, B. E., Durstenfeld, A. & Roehm, P. C. Viral causes of hearing loss: a review for hearing health professionals. Trends Hearing 18, 233121651454136 (2014).
Google Scholar
Perny, M. et al. Streptococcus pneumoniae-induced ototoxicity in organ of Corti explant cultures. Hear. Res. 350, 100–109 (2017).
Google Scholar
Douglas, S. A., Sanli, H. & Gibson, W. P. R. Meningitis resulting in hearing loss and labyrinthitis ossificans—does the causative organism matter? Cochlear Implants Int. 9, 90–96 (2008).
Google Scholar
Sáez-Llorens, X. & McCracken, G. H. Bacterial meningitis in children. Lancet 361, 2139–2148 (2003).
Google Scholar
Lucas, M. J., Brouwer, M. C. & van de Beek, D. Neurological sequelae of bacterial meningitis. J. Infect. 73, 18–27 (2016).
Google Scholar
Du, Y., Wu, X. & Li, L. Mechanisms of bacterial meningitis-related deafness. Drug Discov. Today Dis. Mech. 3, 115–118 (2006).
Google Scholar
Davis, L. E. Acute bacterial meningitis. Continuum 24, 1264–1283 (2018).
Google Scholar
Kim, J., Vasan, S., Kim, J. H. & Ake, J. A. Current approaches to HIV vaccine development: a narrative review. J. Int. AIDS Soc. 24, e25793 (2021).
Google Scholar
Pattnaik, A., Sahoo, B. R. & Pattnaik, A. K. Current status of Zika virus vaccines: successes and challenges. Vaccines 8, 266 (2020).
Poland, G. A., Ovsyannikova, I. G. & Kennedy, R. B. Zika vaccine development: current status. Mayo Clin. Proc. 94, 2572–2586 (2019).
Google Scholar
Wang, Y., Ling, L., Zhang, Z. & Marin-Lopez, A. Current advances in Zika vaccine development. Vaccines 10, 1816 (2022).
Barbosa, M. H. D. M. et al. Auditory findings associated with Zika virus infection: an integrative review. Braz. J. Otorhinolaryngol. 85, 642–663 (2019).
Google Scholar
Mitsikas, D., Gabrani, C., Giannakou, K. & Lamnisos, D. Intrauterine exposure to Zika virus and hearing loss within the first few years of life: a systematic literature review. Int. J. Pediatr. Otorhinolaryngol. 147, 110801 (2021).
Google Scholar
Adler, S. P. et al. Phase 1 clinical trial of a conditionally replication-defective human cytomegalovirus (CMV) vaccine in CMV-seronegative subjects. J. Infect. Dis. 220, 411–419 (2019).
Google Scholar
World Health Organization. Malaria vaccine: WHO position paper—March 2022. (2022).
Zhao, S. Z. & Mackenzie, I. J. Deafness: malaria as a forgotten cause. Ann. Trop. Paediatr. 31, 1–10 (2011).
Google Scholar
Tada, T. et al. A case of falciparum malaria: acute hearing loss as the initial symptom. J. Infect. Chemother. 23, 56–58 (2017).
Google Scholar
Joshi, A., Gupta, N., Ish, P. & Chakrabarti, S. Cerebral malaria: hear before discharge! Indian J. Crit. Care Med 24, 212–213 (2020).
Google Scholar
Mazur, N. I. et al. Respiratory syncytial virus prevention within reach: the vaccine and monoclonal antibody landscape. Lancet Infect. Dis. 23, e2–e21 (2023).
Google Scholar
Venkatesan, P. & First, R. S. V. vaccine approvals. Lancet Microbe 4, e577 (2023).
Google Scholar
Rovers, M. M., Schilder, A. G. M., Zielhuis, G. A. & Rosenfeld, R. M. Otitis media. Lancet 363, 465–473 (2004).
Google Scholar
Centers for Disease Control and Prevention. Respiratory Syncytial Virus Infection (RSV). (2020).
World Health Organization. World Report on Hearing (World Health Organization, 2021).
Kobayashi, M. et al. WHO consultation on group B Streptococcus vaccine development: report from a meeting held on 27–28 April 2016. Vaccine 37, 7307–7314 (2019).
Google Scholar
Heath, P. T. et al. Group B streptococcus and respiratory syncytial virus immunisation during pregnancy: a landscape analysis. Lancet Infect. Dis. 17, e223–e234 (2017).
Google Scholar
Salami, K., Gouglas, D., Schmaljohn, C., Saville, M. & Tornieporth, N. A review of Lassa fever vaccine candidates. Curr. Opin. Virol. 37, 105–111 (2019).
Google Scholar
World Health Organization. Lassa fever. (2017).
Mateer, E. J., Huang, C., Shehu, N. Y. & Paessler, S. Lassa fever–induced sensorineural hearing loss: A neglected public health and social burden. PLOS Neglected Tropical Dis. 12, e0006187 (2018).
Google Scholar
Ibekwe, T. S. et al. Early-onset sensorineural hearing loss in Lassa fever. Eur. Arch. Oto-Rhino-Laryngol. 268, 197–201 (2011).
Google Scholar
Sharma, A. R., Lee, Y.-H., Nath, S. & Lee, S.-S. Recent developments and strategies of Ebola virus vaccines. Curr. Opin. Pharmacol. 60, 46–53 (2021).
Google Scholar
World Health Organization. WHO prequalifies Ebola vaccine, paving the way for its use in high-risk countries. (2019).
Ficenec, S. C., Schieffelin, J. S. & Emmett, S. D. A review of hearing loss associated with Zika, Ebola, and Lassa fever. Am. J. Trop. Med. Hyg. 101, 484–490 (2019).
Google Scholar
Clark, D. V. et al. Long-term sequelae after Ebola virus disease in Bundibugyo, Uganda: a retrospective cohort study. Lancet Infect. Dis. 15, 905–912 (2015).
Google Scholar
World Health Organization. Ebola virus disease. (2021).
Westerberg, B. D., Atashband, S. & Kozak, F. K. A systematic review of the incidence of sensorineural hearing loss in neonates exposed to Herpes simplex virus (HSV). Int. J. Pediatr. Otorhinolaryngol. 72, 931–937 (2008).
Google Scholar
Rabinstein, A., Jerry, J., Saraf–Lavi, E., Sklar, E. & Bradley, W. G. Sudden sensorineural hearing loss associated with herpes simplex virus type 1 infection. Neurology 56, 571 (2001).
Google Scholar
Sohal, K., Moshy, J., Owibingire, S. & Shuaibu, I. Hearing loss in children: a review of literature. J. Med. Sci. 40, 149–161 (2020).
Google Scholar
Johnston, C., Gottlieb, S. L. & Wald, A. Status of vaccine research and development of vaccines for herpes simplex virus. Vaccine 34, 2948–2952 (2016).
Google Scholar
Sandgren, K. J., Truong, N. R., Smith, J. B., Bertram, K. & Cunningham, A. L. Vaccines for herpes simplex: recent progress driven by viral and adjuvant immunology. Methods Mol. Biol. 2060, 31–56 (2020).
Google Scholar
Piras, F., Plitnick, L. M., Berglund, P., Bernard, M. C. & Desert, P. Nonclinical safety evaluation of two vaccine candidates for herpes simplex virus type 2 to support combined administration in humans. J. Appl. Toxicol. 43, 534–556 (2023).
Google Scholar
Chentoufi, A. A., et al. Combinatorial herpes simplex vaccine strategies: from bedside to bench and back. Front. Immunol. 13, 849515 (2022).
link