Agriculture is one of many domains where a (r)evolution is essential to meet the challenge of pressing and variable environmental constraints, as well as increasing demographic pressure. Several paradigms initiated the transformation of our food and agricultural systems, such as agroecology, which proposes to apply ecological principles in agriculture1. Most changes are driven by the need to efficiently cope with the constraints surrounding agricultural activities: resources are finite and all ecosystems are dynamic and interconnected. Since the green revolution initiated in the 1960s, research in agriculture has been compartmentalized, with the dominant idea that there exists a human-made solution to every environmental constraint, mainly relying on the use of synthetic molecules, the artificialization of the production environments, and with plant and animal breeding goals focused only on production performances. Unfortunately, the broad impact of these solutions on ecosystem health, including the food systems and human health, has been largely underestimated2. Indirectly, this eradication strategy created a new set of constraints, probably stronger3,4,5. Fundamentally, the paradigm shifting toward the development of control methods that aim to reach favorable equilibrium for agricultural production is a necessity. Productivity could not be the only variable while designing new agricultural practices; ecosystems, animal, and human health must be considered too, requiring collaborative, transdisciplinary, and intersectoral approaches in accordance with the One Health concept6,7.
Gastro-Intestinal Nematodes (GIN) are a major threat for ruminants at pasture worldwide8. It is a meaningful example of how the management strategies shift from total eradication based on anthelmintic (AH) chemical treatments to management of the GIN populations in order to maintain a sustainable equilibrium favorable for animal production9. The 1960-1980s was the golden era for AH treatments10 and other strategies were ignored. Necessarily, the systematic use of AH created a selection pressure driving the apparition of AH resistant GIN strains worldwide. Nowadays, the rise of AH resistance is one major concern of small ruminant husbandry8,11. It is now accepted by most parasitologists that integrated GIN management is the only sustainable solution9,12,13,14,15, however clear and general guidelines are still needed, to change management in practice, which still often relies on systematic AH use.
Formally, the current challenge for health management in agriculture is to consider a set of broader, multidisciplinary objectives and constraints (e.g. ecosystem, animal and human health, biodiversity, productivity), under great uncertainty (e.g. climate change, availability of curative treatments and resources). Rationalizing decisions, with the available knowledge and expertise, could help in reaching this goal16.
In this article, we present a mathematical model to balance the impact of prevention and control in disease management. This simple framework allows to highlight and discuss the key parameters of a disease management problem, making it particularly useful for engaging stakeholders with diverse backgrounds. The model facilitates the classification of potential management actions and explores their relationships to the disease from an economic perspective. The optimality results guide decision-making by identifying which actions are worth implementing, the ideal balance between prevention and control, the required level of efficacy, and the associated costs. It is suited to support generic discussions about a disease management problem, at a global scale. Although it is a small step considering the challenge of health management listed earlier, generic frameworks are needed to help the adoption of alternatives and more integrated methods in practice.
This framework differs from previous studies in its capacity to incorporate simultaneously the effects of prevention and control strategies, allowing discussion of the optimal trade-off between both. It is illustrated in a practical case, the management of gastrointestinal nematodes, a worldwide sanitary constraint for small ruminants, where more integrated management is needed to prevent drug resistance. The framework is used to discuss the economic feasibility of alternative methods, to help in their use and development in practice.
Related works
The use of mathematical models to optimize decisions is well-suited to various areas of animal and human health management16,18,19,20, as well as ecosystem management21,22,23. Quantifying the impact of prevention is often challenging because the outcomes of preventive actions are not always straightforward to estimate20.
In human health, cost-effectiveness or cost-utility ratios are commonly used. These metrics represent the cost of a preventive action divided by an estimate of health outcomes, such as quality-adjusted life years (QALYs)24,25, which takes into account both life expectancy and quality of life. However, this approach is difficult to transfer to the animal health context, where quantitative metrics for animal welfare remain underdeveloped. Nevertheless, the underlying principle remains the same: using data to inform decisions by balancing the relative benefits of management actions against their costs.
The use of mathematical models for disease-control decision-making has been reviewed in16 for livestock or more recently in17,32 for human health. In the former, they classified quantitative modeling into two categories: statistical and/or epidemiological models, and economic models. Economic models are further divided into five categories: (i) mathematical programming, (ii) network analysis, (iii) decision analysis, (iv) simulation, and (v) cost-benefit analysis. Most modeling works on disease management strategies fall under the fourth category, simulation models27,28,29. In these cases, decisions are not optimized per se; rather, the objective is to estimate the potential effects and utility of a given management strategy. A model of disease dynamics is first developed, and then the costs and benefits of the strategy are estimated by running the model under the proposed management scenario. This approach is particularly suitable for evaluating whether a management strategy, such as vaccination, is worthwhile28, but it does not necessarily identify the optimal strategy among all possible alternatives.
The work presented in this article belongs to the fifth category30,31,32, as it frames the challenge of identifying an optimal disease-control strategy as a resource allocation problem. Utility is based on estimates of the benefits and costs associated with the management strategy. We followed the fundamental steps recommended for developing quantitative models to aid decision-making16,26. Our process involved developing a conceptual model, formulating a mathematical representation of the decision problem, and designing an algorithmic procedure to optimize the decision. It is an extension of the works presented in31,33, with the particular case of a unique population and where we explicitly propose a utility function in the case of gastrointestinal nematodes in small ruminants. A major difference also comes from the fact that we both include prevention and control simultaneously, unlike in the original work, where only prevention was included.
Our approach is particularly innovative in the field of animal health management, as it focuses on identifying an optimal disease management strategy through an optimization procedure. This approach is more common in health economic34,35 and ecosystem management/quantitative ecology, where mathematical modeling and optimization have been widely used to address issues such as managing invasive species or conserving threatened populations21,22,23. This work draws particular inspiration from21, where the objective was to determine an invasive species surveillance strategy, while minimizing management costs.
Framing a disease management problem
Mathematical models offer a valuable tool for analyzing the impact of decisions on dynamic systems. Here we provide a simple framework capturing the key elements of a disease affecting a farming production system. Management strategies are compared in terms of farmer’s profit, denoted G, a common variable to compare the effect of management strategies. For examples, G could be the gain after selling an animal product (e.g. meat, milk, eggs) or after harvesting the crop field. It could also be based on a biodiversity or a durability index, or on ecosystem services in other applications. One constraint is to be able to compare G with the management effort, and thus need to be on the same currency, which could be difficult. G is a random variable, meaning that at the start of production, its value is unknown and will only be determined at the end when the production is sold.
It is however useful to discuss management decisions before implementing them, considering the expected value of G, based on the chosen strategy. In essence, defining a probability distribution for the variable G allows for a more informed decision-making process.
We considered a generic situation applicable to both plant and animal production system. We considered two scenarios, depending either a sanitary problem occurred or not, with a probability \(p_m\). When no sanitary problem occurred, with probability \(1-p_m\), the farmer’s gain is optimal, denoted \(G^*\), indicating that production remains unaffected by the sanitary problem. When stress occurs, with a probability of \(p_m\), some losses are expected, i.e. \(G
Two scenarios are considered after the occurence of the health issue. Firstly, production may cease entirely, resulting in complete loss. This occurs, for example, when an animal dies. Let \(p_s\) denote the survival probability, indicating the likelihood of the animal or production system surviving the health stress. Thus, with a probability of \(1-p_s\), \(G=0\), signifying no gain is derived from the production system.
Secondly, the production system may not cease completely but rather be affected by the health stress. Consequently, only a proportion \(p_r\) of the optimal gain will be obtained from the production.
To summarize, the variable G is equal to \(G^*\), with probability \(1-p_m\), is equal to \(p_r G^*\) with probability \(p_m p_s\), and to 0 with probability \(p_m (1-p_s)\).
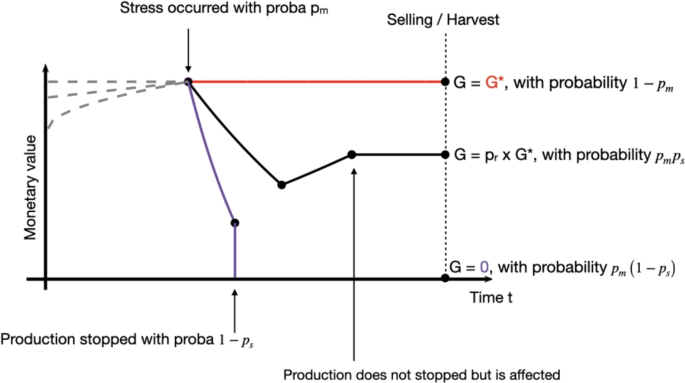
This framework aims to describe the disease management problem using only four variables: the optimal gain \(G^*\), the frequency of stress occurrence \(p_m\), the survival probability of the production system in the event of a health stress \(p_s\), and the system’s recovery capacity after the stress occurs \(p_r\). The framework is illustrated in Fig. 1.
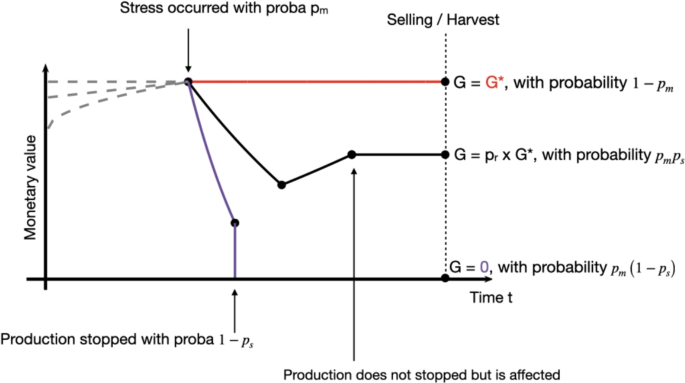
Illustration of the disease management framework: possible trajectories of the monetary value (MV) of the system over time. In normal situation, MV is an increasing function, as illustrated by the gray dotted lines. In a context of animal sold for meet for example, the monetary value mostly depends on the animal weight, which increases with time. Three scenarios are considered: (red) no sanitary stress occurred, with probability \(1-p_m\) and the optimal gain \(G^*\) is received. (black) the stress occurred, the production is affected, but did not stopped, with probability \(p_s\), and only \(p_r G\) is received. (purple) the stress occurred and totally stopped the production, no gain are received. For this last two scenarios, it can be for example the case of a fruit tree. A disease happened and some fruits are lost (decreasing monetary value). The farmer then applied some treatment. The purple line indicates a case where the treatment does not work, all the fruits are lost. The black line is another possible scenario, the treatment worked, although the lost fruits will not be sold and thus represent losses (i.e. \(G
Management strategy
The decision problem involves examining various management options and their impact on the final gain G.
Generally, there are two classes of management options: prevention and control. Prevention encompasses any management strategies implemented at the beginning of the production system, regardless of whether a health stress occurs or not. On the other hand, control strategies are only implemented upon detecting a stress.
Initially, control may seem like the optimal option because investments are only made in response to a health stress. Conversely, investments in prevention are lost if no stress occurs. This article aims to mathematically derive the situations in which it is optimal to use one or both of these management options and to discuss the practical implications of these decisions.
Prevention encompasses actions aimed at limiting the occurrence of health stress, thereby reducing \(p_m\). It also includes measures to detect stress as early as possible, thereby increasing the survival probability \(p_s\) or enhancing the recovery capacity \(p_r\). Control, on the other hand, does not affect the frequency of stress occurrence, but only \(p_s\) and \(p_r\).
To compare management options, it is essential to define a criterion. A natural criterion is based on the final gain G, and we propose to use the expected loss due to the health stress, denoted as \(V_0\):
$$\begin{aligned} V_0= & \textbf{E}\left[ G^*-G\right] =p_m\times \left( G^*\left( 1-p_r*p_s\right) \right) ,\nonumber \\= & p_m\times p_f. \end{aligned}$$
(1)
\(\textbf{E}\) is the notation for the mathematical expectation of a random variable. \(V_0\) represents the anticipated loss resulting from stress when no management action is taken, or in other words, it’s the expected disparity between the optimal benefit \(G^*\) and the farmer’s actual benefit G. \(V_0\) is the initial value of the management problem, and \(p_f=G^*\left( 1-p_r*p_s\right)\) denotes the expected ultimate loss in case of stress occurrence.
Integrated Health Management (IHM) strategy aims to reduce \(V_0\) through various means.
A management strategy is characterized by a combination of prevention and control efforts, denoted as \(\left( x_p,x_c\right)\), along with their corresponding efficacy per unit effort \(\left( \lambda _p,\lambda _c\right)\). The value of a management strategy is defined as:
$$\begin{aligned} V= & V_0\times \exp \left( -\lambda _p x_p-\lambda _c x_c\right) + x_p + p_m x_c. \end{aligned}$$
(2)
The expression \(V_0\exp \left( -\lambda _p x_p-\lambda _c x_c\right)\) represents the updated expected loss resulting from the stress, considering the implementation of certain management efforts \(x_p\) and \(x_c\).
It’s important to note:
$$\exp \left( -\lambda _p x_p-\lambda _c x_c\right) =\exp \left( -\lambda _p x_p\right) \exp \left( -\lambda _c x_c\right) .$$
And the term \(\Delta _p=\exp \left( -\lambda _p x_p\right)\) represents the expected reduction attributed to prevention, while \(\Delta _c=\exp \left( -\lambda _c x_c\right)\) signifies the expected reduction attributed to control efforts. The expression \(x_p+p_m x_c\) denotes the anticipated management cost, or simply, the expected loss solely due to management expenses. Since \(x_c\) is associated only with the occurrence of stress, its anticipated cost is scaled by the probability of stress, \(p_m\). The value V represents the anticipated loss post-management, encompassing both stress-related losses and management expenses. Utilizing V enables the comparison of different strategies, with the objective of minimizing this value.
The efficacies \(\lambda _p\) and \(\lambda _c\) quantify the rate at which the expected loss \(V_0\) decreases for a given amount of prevention \(x_p\) and control efforts \(x_c\). Efficacy is used to model the fact that different strategy might not be as efficient than other. Or in other words, that for the same amount of effort, \(x_c\) or \(x_p\), we might not have the same loss reduction for all possible strategies. We used an exponential relationship to model diminishing marginal returns, implying that a small amount initially gives the biggest payoff in cost reduction21. Alternative increasing functions, such as logit or piecewise linear functions, could also be employed. However, they might introduce complexity in determining the optimal solution, although computing numerical solutions might be feasible.
Optimal integrated health management strategy
The optimal integrated health management (IHM) strategy is the one allowing the optimal trade-off between loss reduction and management costs, i.e minimizes V. Then, the disease management problem can be reformulated as an optimization problem to determine the optimal IHM strategies based on the problem’s parameters.
The following rules of thumb can be derived from the computation of the optimal management strategy.
Management decisions should primarily depend on our ability to achieve measurable impacts on the situation, which is determined by the efficacy of our management actions. Efficacy refers to the relative value of the effects of our actions (decreasing \(V_0\)) compared to their costs.
An expensive management action that has little effect on the sanitary problem, i.e., on stress occurrence frequency \(p_m\), individual survival when the stress occurs \(p_s\), or recovery capacity \(p_r\), is considered of low interest.
For prevention strategies, efficacy should be compared to the expected loss of the sanitary problem (\(p_m \times p_f\)). Prevention becomes worthwhile, i.e., it creates a positive trade-off between management cost and stress expected loss, when \(\lambda _p > \frac{1}{p_m \times p_f}\).
Control efficacy should be compared to the loss when the stress occurs (\(p_f\)). Control measures are considered worthwhile when \(\lambda _c > \frac{1}{p_f}\).
If the efficacies fall below these thresholds, it is not worth implementing any management action, as the investment does not justify the expected reward.
On the other hand, when the control efficacy exceeds \(p_f\) and is higher than the prevention efficacy, control measures should always be prioritized.
If we achieve better results with the same investment using control measures, it is not justified to allocate resources to prevention efforts. To be the optimal strategy, the efficacy of prevention measures should significantly surpass that of control measures.
Expressing \(\lambda _c = \lambda _p – \delta\), where \(\delta > 0\) represents the difference in efficacy between prevention and control, it can be shown that \(\delta\) must exceed \(\left( 1-p_m\right) \lambda _p\) for prevention to be the optimal choice.
The term \(\left( 1-p_m\right) \lambda _p\) approaches zero as \(p_m\) approaches 1. In other words, the requirement for additional prevention efficacy relative to control decreases as the frequency of the sanitary problem increases.
The optimal effort allocation is summarized in Fig. 2.
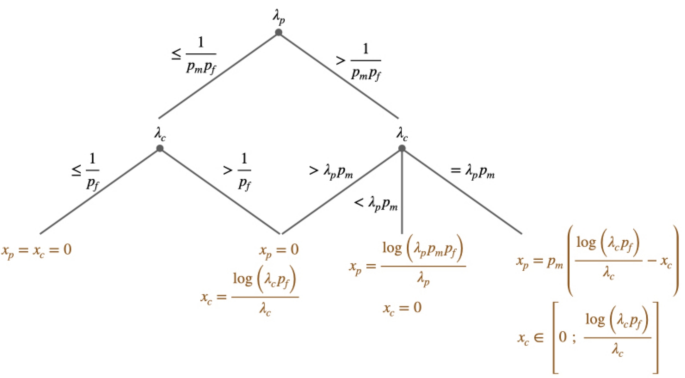
Decision tree summarizing the optimal effort allocation. Nodes represent the efficacy parameters and each branch their possible values. Final nodes are the optimal effort allocation. For example, the left part of the tree indicates that when \(\lambda _p\le \frac{1}{p_m p_f}\) and \(\lambda _c\le \frac{1}{p_f}\), then it is optimal to do nothing, i.e. \(x^*_p=x^*_c=0\).
Several examples of the V function as well as optimal and sub-optimal management strategies are provided Fig. 3.
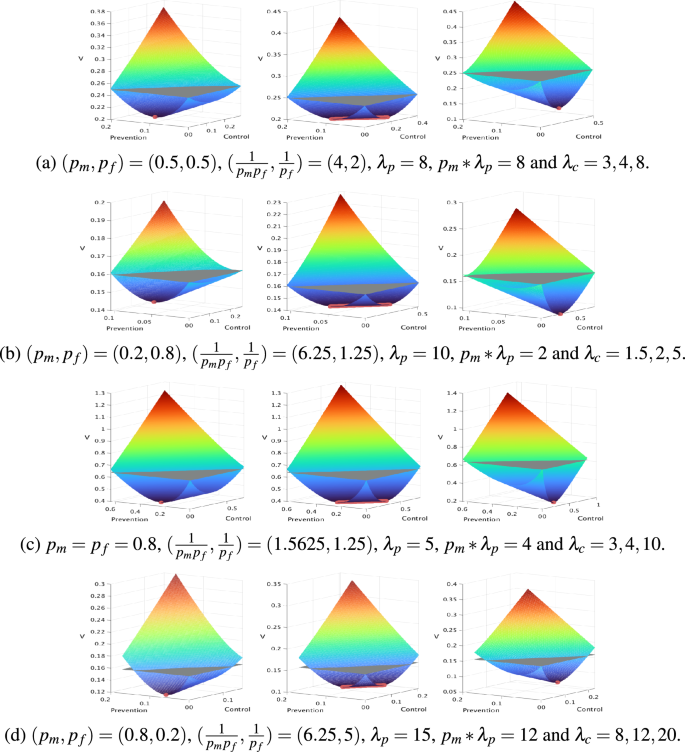
Examples of the V function. For each example, the function V is displayed in color, while the plane \(z=V_0\) is displayed in gray. The x and y-axis are the amount of prevention and control efforts. Efforts are normalized with \(G^*\). The z-axis is the value V of a management strategy, for the corresponding amount of prevention and control. All the parameters (prevention and control costs and V) are normalized by \(G^*\). When V is under the plane, the trade-off between management effect and cost is positive. Each row, (a), (b), (c), (d) of figures gives examples when \(\lambda _c > \frac{1}{p_f}\) and \(\lambda _p>\frac{1}{p_m p_f}\), i.e. when a positive trade-off can be expected. Each figure gives three examples, with the same value of \(p_m\), \(p_f\) and \(\lambda _p\), but various values of \(\lambda _c\): under (left), equal (center) or above (right) \(p_m*\lambda _p\). Finally, the optimal management efforts are displayed with red points on the V function. For each row, the left figure is for \(\lambda _c
link